Advertisements
Advertisements
Question
Write the mechanism of the following reaction:
\[\ce{{n}BuBr + KCN ->[EtOH-H2O] {n}BuCN}\]
Advertisements
Solution 1
This reaction is a first-order nucleophilic substitution (SN1). The mechanism can be stated as:
Step 1: Generation of nucleophile:
\[\ce{KCN ->[EtOH-H2O]K+ + \overset{—}{C} ≡ N}\]
Step 2: Nucleophilic attack and formation of transition state:
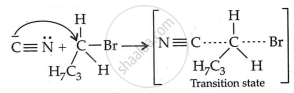
Step 3: Generation of product:
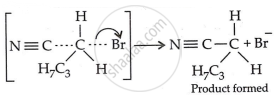
Therefore, we have
\[\ce{CH3 - CH2 - CH2 - CH2 - Br + KCN ->[EtOH/H2O]CH3CH2CH2CH2CN + KBr}\]
Solution 2
KCN is the resonating hybrid of the following structures:
\[\ce{K^+ [^- :C ≡ N: ↔ :C = \overset{\bullet\bullet}{N} :^-]}\]
Therefore, CN⁻ acts as an ambident nucleophile. It can attack the carbon atom of the C-Br bond in n-BuBr through either the carbon (C) or nitrogen (N) atom. Since the C-N bond is weaker than the C-C bond, the attack occurs at the carbon atom, leading to the formation of n-butyl cyanide.
\[\ce{K^+CN^- + \underset{n-butyl bromide}{CH3CH2CH2\overset{δ+}{C}H2 - \overset{δ-}{B}r} -> \underset{n-butyl cyanide}{CH3CH2CH2CH2CN} + KBr}\]
Notes
Students can refer to the provided solutions based on their preferred marks.
APPEARS IN
RELATED QUESTIONS
Discuss the mechanism of alkaline hydrolysis of bromomethane.
Which would undergo SN2 reaction faster in the following pair and why ?

Which compound in the following pair will react faster in SN2 reaction with OH−?
CH3Br or CH3I
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH2OH + SOCl2 ->}\]
Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolysed by aqueous KOH.
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
Which compound in the following pair reacts faster in SN2 reaction with OH–?
- CH3Br or CH3
- CH3Cl, (CH3)3CCl
An important chemical method to resolve a racemic mixture makes use of the formation of ______.
Among the following, the dissociation constant is highest for:
Assertion: KCN reacts with methyl chloride to give methyl isocyanide.
Reason: CN– is an ambident nucleophile.
Which reagent will you use for the following reaction?
\[\ce{CH3CH2CH2CH3 -> CH3CH2CH2CH2Cl + CH3CH2CHClCH3}\]
CCl4 is insoluble in water because:-
Among the following compounds I - IV, which one forms a yellow precipitate on reacting sequentially with (i) NaOH (ii) dil. HNO3 (iii) AgNO3?
 |
 |
 |
 |
| I | II | III | IV |
Discuss SN2 mechanism of methyl bromide using aqueous KOH.
Acetic anhydride from acetic acid
Explain why Grignard reagents should be prepared under anhydrous conditions.
 HCI, Major product ______.
HCI, Major product ______.
Consider the reactions,
(i) \[\begin{array}{cc}
\phantom{}\ce{\underset{}{(CH3)2CH - CH2Br} ->[C2H5OH] \underset{}{(CH3)2CH - CH2OC2H5 + HBr}}\\
\end{array}\]
(ii) \[\begin{array}{cc}
\phantom{}\ce{\underset{}{(CH3)2CH - CH2Br} ->[C2H5O-] \underset{}{(CH3)2CH - CH2OC2H5 + Br-}}\\
\end{array}\]
The mechanisms of reactions (i) and (ii) are respectively:
Assertion (A):  undergoes SN2 reactions faster than
undergoes SN2 reactions faster than  .
.
Reason (R): Iodine is a better leaving group because of its large size.
In the light of the above statements, choose the correct answer from the options given below:
