Advertisements
Advertisements
Question
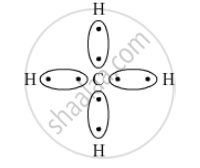
What type of bonds are present in CH4 molecule? Draw their electron-dot structures.
Advertisements
Solution
Covalent bond is the chemical bond present in CH4 as a carbon atom shares its electrons with four hydrogen atoms.
The electron-dot structure is:
APPEARS IN
RELATED QUESTIONS
Name the following:
The property of element by virtue of which atoms of the element can link to each other in the form of a long chain or ring structure.
What type of bonds are present in water molecule? Draw the electron-dot structure of water (H2O).
What is buckminsterfullerene?
will CS2 conduct electricity or not?
give reason for your choice.
Using electron-dot diagrams which show only the outermost shell electrons, show how a molecule of oxygen, O2, is formed from two oxygen atoms. What name is given to this type of bonding? (At. No. of oxygen = 8)
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
The atomic numbers of four elements P, Q, R and S are 6, 10, 12 and 17 respectively. Which two elements can combine to form a covalent compound?
(a) P and R
(b) Q and S
(c) P and S
(d) R and S
One of the following compounds is not ionic in nature. This compound is:
(a) Lithium chloride
(b) Ammonium chloride
(c) Calcium chloride
(d) Carbon tetrachloride
A covalent molecule having a double bond between its atoms is:
(a) Hydrogen
(b) Oxygen
(c) water
(d) ammonia
State the type of bonding in the following molecule.
Ammonium ion
Explain the following term with example.
Alkane
Fill in the blank with correct word from the bracket.
Melting and boiling points of covalent compounds are generally ______ (low, high).
What are the term defined below?
A bond formed by share pair of electrons, each bonding atom contributing one electron to the pair.
What do you understand by dipole (polar) molecule?
Fill in the blank and rewrite the completed statement:
Covalent compounds are generally soluble in _________ solvents.
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Cation is formed when ______.
An element A is soft and can be cut with a knife. This is very reactive to air and cannot be kept open in air. It reacts vigorously with water. Identify the element from the following
Oils on treating with hydrogen in the presence of palladium or nickel catalyst form fats. This is an example of
______ is an example of a covalent compound having a high melting point.
