Advertisements
Advertisements
प्रश्न
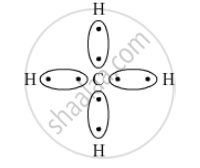
What type of bonds are present in CH4 molecule? Draw their electron-dot structures.
Advertisements
उत्तर
Covalent bond is the chemical bond present in CH4 as a carbon atom shares its electrons with four hydrogen atoms.
The electron-dot structure is:
APPEARS IN
संबंधित प्रश्न
What type of chemical bonds are formed by carbon? Why?
How buckminsterfullerene is it related to diamond and graphite?
Why is graphite used for making dry cell electrodes but diamond is not?
Name one covalent compound containing chlorine.
How will you find out which of the water soluble compound A or B is ionic?
What type of bonding would you expect between Hydrogen and Chlorine?
Explain why, graphite can be used as a lubricant but diamond cannot.
State any two uses of diamond.
Describe the structure of graphite with the help of a labelled diagram.
An element E exists in three allotropic forms A, B and C. In allotrope A, the atoms of element E are joined to form spherical molecules. In allotrope B, each atom of element E is surrounded by three other E atoms to form a sheet like structure. In allotrope C, each atom of element E is surrounded by four other E atoms to form a rigid structure.
(a) Name the element E.
(b) What is allotrope A.
(c) What is allotrope B?
(d) What is allotrope C?
(e) Which allotrope is used in making jewellery?
(f) Which allotrope is used in making anode of a dry cell?
What is the difference between ionic compounds and covalent compounds?
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Water
Give examples for the following:
Two gaseous non polar compounds.
Explain the structure of Ammonium ion.
Explain the following:
Covalent compounds are generally gases or liquids or soft solids.
What is the term defined below?
A bond formed by a shared pair of electrons, each bonding atom contributing one electron to the pair.
State the type of bond formed when the combining atom has zero E.N. difference.
What is the difference between :
Ionic compounds and covalent compounds
List two differences between the properties exhibited by covalent compounds and ionic compounds.
Mineral acids are stronger acids than carboxylic acids because
- mineral acids are completely ionised
- carboxylic acids are completely ionised
- mineral acids are partially ionised
- carboxylic acids are partially ionised
