Advertisements
Advertisements
प्रश्न
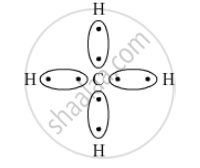
What type of bonds are present in CH4 molecule? Draw their electron-dot structures.
Advertisements
उत्तर
Covalent bond is the chemical bond present in CH4 as a carbon atom shares its electrons with four hydrogen atoms.
The electron-dot structure is:
APPEARS IN
संबंधित प्रश्न
Name the type of bonds formed in ionic compounds and in the compounds formed by carbon.
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many hexagons of carbon atoms are present in one molecule of buckminsterfullerene?
What would be the electron-dot structure of carbon dioxide which has the formula CO2?
What do you call the compounds having the same molecular formula but different structural arrangements of atoms?
What type of bonds are present in CO2 molecule? Draw their electron-dot structures.
Which inert gas does the Cl atom in HCl resemble in electron arrangement?
The electronic configurations of three elements X, Y and Z are as follows:
| X | 2, 4 |
| Y | 2, 7 |
| Z | 2, 1 |
(a) Which two elements will combine to form an ionic compound?
(b) Which two elements will react to form a covalent compound?
Give reasons for your choice.
Which element exhibits the property of catenation to maximum extent and why?
Choose and write the correct option.
Which type of carbon-carbon bonds are present in Vanaspati ghee?
Give reason as to why hydrogen chloride can be termed as a polar covalent compound.
State the type of bonding in the following molecule.
Water
Taking MgCl2 as an electrovalent compound, CCl4 as a covalent compound, give four difference between electrovalent and covalent compounds
Number of valence electrons in a carbon atom is _______.
Write an Explanation.
Alkene
Write a short note.
Aromatic hydrocarbons
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc}\phantom{......}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}\\ \phantom{.....}|\phantom{....}|\phantom{....}|\\ \ce{H - C - C = C}\\\phantom{.....}|\phantom{.........}|\\ \phantom{.....}\ce{H}\phantom{........}\ce{H}\end{array}\] |
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc} \phantom{.........}\ce{H}\\ \phantom{.........}|\\ \ce{H - C ≡ C - C - H}\\ \phantom{.........}|\\ \phantom{.........}\ce{H} \end{array}\] |
The number of pi-bonds present in benzoic acid molecule are ______.
Statement (A): Covalent compounds are bad conductors of electricity.
Reason (B): Covalent compounds contain charged particles (ions)
Non-polar covalent compounds are ______ conductors of heat and electricity.
