Advertisements
Advertisements
प्रश्न
What type of bonds are present in CO2 molecule? Draw their electron-dot structures.
Advertisements
उत्तर
Covalent bond is the bond present in O2 as one oxygen atom shares its electrons with another oxygen atom.
The electron-dot structure is:
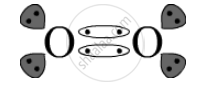
APPEARS IN
संबंधित प्रश्न
Why covalent compounds are different from ionic compounds?
Give reason why carbon compounds are generally poor conductors of electricity.
Give appropriate scientific reasons for Carbon tetrachloride does not conduct electricity.
State one major difference between covalent and ionic bonds and give one example each of covalent and ionic compounds.
Which inert gas electron configuration do the Cl atoms in Cl2 molecule resemble? What is this electron configuration?
State any two uses of graphite.
The solution of one of the following compounds will not conduct electricity. This compounds is:
(a) NaCl
(b) CCl4
(c) MgCl2
(d) CaCl2
The number of covalent bonds in pentane (molecular formula C5H12) is:
The electronic configurations of three elements X, Y and Z are as follows:
| X | 2, 4 |
| Y | 2, 7 |
| Z | 2, 1 |
(a) Which two elements will combine to form an ionic compound?
(b) Which two elements will react to form a covalent compound?
Give reasons for your choice.
Draw an electron dot diagram for the formation of the following. State the type of bonding present in them.
Hydroxyl ion
Draw all possible structural formulae of compound from their molecular formula given below.
C3H8
Explain the following term with example.
Covalent bond
Explain the following briefly:
Cl2 is a non polar molecule, while HCl is a polar molecule.
State the type of bond formed when the combining atom has zero E.N. difference.
State the type of bond formed, and draw Lewis structure of water.
Name two compounds that are covalent when taken pure but produce ions when dissolved in water.
Name gas with a rotten egg smell.
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Which of the following statements are correct for carbon compounds?
- Most carbon compounds are good conductors of electricity.
- Most carbon compounds are poor conductors of electricity.
- Force of attraction between molecules of carbon compounds is not very strong.
- Force of attraction between molecules of carbon compounds is very strong.
Two carbon atoms can always form one or two covalent bonds.
