Advertisements
Advertisements
प्रश्न
What type of bonds are present in CO2 molecule? Draw their electron-dot structures.
Advertisements
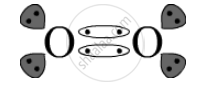
उत्तर
Covalent bond is the bond present in O2 as one oxygen atom shares its electrons with another oxygen atom.
The electron-dot structure is:
APPEARS IN
संबंधित प्रश्न
State the reason to explain why covalent compounds "are bad conductors of electricity".
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many pentagons of carbon atoms are present in one molecule of buckminsterfullerene?
Give one example of a molecule containing a double covalent bond
Fill in the blanks in the following sentence:
In forming oxygen molecule, .............. electrons are shared by each atom of oxygen.
What is the atomic number of carbon. Write its electronic configuration.
Explain why covalent compounds have generally high melting points?
Give the formula of the compound that would be formed by the combination of the following pair of elements:
K and H
Explain why, diamond is hard while graphite is soft (though both are made of carbon atoms).
What is graphite?
A solid element X has four electrons in the outermost shell of its atom. An allotrope Y of this element is used as a dry lubricant in machinery and also in making pencil leads.
(a) What is element X?
(b) Name the allotrope Y.
(c) State whether allotrope Y is a good conductor or non-conductor of electricity.
(d) Name one use of allotrope Y (other than lubrication and pencil leads)
(e) Name two other allotropes of element X.
What do you understand by polar covalent compounds?
Draw all possile structural formulae of compound from their molecular formula given below.
C3H4
Explain the Structural isomerism term with example.
Explain the following briefly:
Sodium chloride dissolves in water but carbon tetra chloride is insoluble in water.
Give examples for the following:
Two gaseous non polar compounds.
Draw an electron dot diagram to show the formation of the following compound.
Magnesium chloride [ H=1, C=6, Mg=12, Cl=17].
Explain the following:
Polar covalent compounds conduct electricity?
In the formation of electrovalent compounds, electrons are transferred from one element to another. How are electrons involved in the formation of a covalent compound?
Discuss in brief about the properties of coordinate covalent compounds.
The number of pi-bonds present in benzoic acid molecule are ______.
