Advertisements
Advertisements
Question
What type of bonds are present in CO2 molecule? Draw their electron-dot structures.
Advertisements
Solution
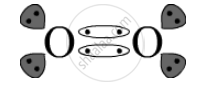
Covalent bond is the bond present in O2 as one oxygen atom shares its electrons with another oxygen atom.
The electron-dot structure is:
APPEARS IN
RELATED QUESTIONS
Fill in the blanks in the following sentence:
In forming oxygen molecule, .............. electrons are shared by each atom of oxygen.
Give two general properties of ionic compounds and two those of covalent compounds.
Draw the electron-dot structure of CO2 compound and state the type of bonding.
Explain why, diamond can be used in rock drilling equipment but graphite cannot.
Give two example in following case:
Gaseous polar compounds
Give two example in following case:
Gaseous non polar compounds
Observe the straight chain hydrocarbons given below and answer the following
questions :
i) Which of the straight chain compounds from A and B is saturated and unsaturated straight chains?
ii) Name these straight chains.
iii) Write their chemical formulae and number of – CH2 - units.

Choose the correct answer from the options given below
Condition favorable for formation of a covalent bond is
Name a neutral covalent molecule which contains one lone pair of electrons.
Give reason as to why hydrogen chloride can be termed as a polar covalent compound.
Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the options A, B, C and D given below:
The type of bonding in X will be ______.
Electrons are getting added to an element Y:
which electrode will Y migrate to during the process of electrolysis?
Explain why carbon tetrachloride does not dissolve in water.
Element A has 2 electrons in its M shell. Element B has atomic number 7.
If B is a diatomic gas, write the equation for the direct combination of A and B to form a compound.
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Discuss in brief about the properties of coordinate covalent compounds.
Carbon forms four covalent bonds by sharing its four valence electrons with four univalent atoms, e.g., hydrogen. After the formation of four bonds, carbon attains the electronic configuration of ______.
Which of the following statements are usually correct for carbon compounds? These
- are good conductors of electricity
- are poor conductors of electricity
- have strong forces of attraction between their molecules
- do not have strong forces of attraction between their molecules.
Write notes on the characteristics of covalent compounds.
