Advertisements
Advertisements
Question
Three organic compounds A, B and C have the following molecular formulae: C4H10O
Which compound contains a carboxyl group? Write its name and structural formula.
Advertisements
Solution
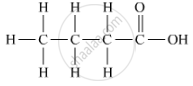
The organic compound 'A' contains a carboxyl group. The name of the acid is butanoic acid, and its formula is C3H7COOH. Its structural formula is as follows:
APPEARS IN
RELATED QUESTIONS
In order to study saponification reaction, we first prepare 20% solution of sodium hydroxide. If we record the temperature of this solution just after adding sodium hydroxide flakes to water and also test its nature using litmus, it may be concluded that the process of making this solution is
(A) exothermic and the solution is alkaline
(B) endothermic and the solution is alkaline
(C) endothermic and the solution is acidic
(D) exothermic and the solution is acidic
Complete the following chemical equations: C2H5OH+O2→
Write the formulae of methanoic acid.
propanol into propanoic acid?
Name the process in each case and write the equations of the reactions involved.
What is an oxidising agent?
Name two oxidising agents which can oxidise ethanol to ethanoic acid.
Name the reducing agent used to convert acetic acid into ethanol.
If you take a pinch of sodium hydrogen carbonate powder in a test-tube and add drop-by-drop acetic acid to it, what would you observe immediately? List any two main observations.
A student is studying the properties of acetic acid in his school laboratory. List two physical and two chemical properties which he must observe and note in his record book.
Why is Soda lime used not only NaOH?
What is the main constituent of vinegar?
State how the following conversions can be carried out:
Ethyl alcohol to Ethane
When sodium hydrogen carbonate is added to ethanoic acid, a gas evolves. Consider the following statements about the gas evolved.
- It turns lime water milky.
- It is evolved with brisk effervescence.
- It has a smell of burning sulfur.
- It is also a by-product of respiration.
Observe the figure and write the answers to the following questions.

- Write the name of the reaction shown in the following figure.
- Write the above chemical reaction in the form of a balanced equation.
- Write the name of the product produced in the above reaction, write a use.
- Write the name of the catalyst used in the above reaction.
Write the answers to the questions by observing the following figure.

- Write the chemical reaction shown in the figure above in the form of a balanced equation.
- Write the name of the gas coming out of the large test tube in the above chemical reaction.
- Why do small bubbles appear in the small test tube?
- What is the change in colour of lime water?
When ethanoic acid is treated with NaHCO^ the gas evolved is ______.
Write an equation showing saponification.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
