Advertisements
Advertisements
प्रश्न
Three organic compounds A, B and C have the following molecular formulae: C4H10O
Which compound contains a carboxyl group? Write its name and structural formula.
Advertisements
उत्तर
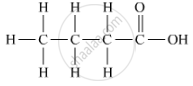
The organic compound 'A' contains a carboxyl group. The name of the acid is butanoic acid, and its formula is C3H7COOH. Its structural formula is as follows:
APPEARS IN
संबंधित प्रश्न
Write three different chemical reactions showing the conversion of ethanoic acid to sodium ethanoate. Write balanced chemical equation in each case. Write the name of the reactants and the products other ethanoic acid and sodium ethanoate in each case.
When you add about 2 ml of acetic acid to a test tube containing an equal amount of distilled water and leave the test tube to settle after shaking its contents, what will you observe in the test tube after about 5 minutes?
(A) A white precipitate settling at its bottom
(B) A clear colourless solution
(C) A layer of water over the layer of acetic acid
(D) A layer of acetic acid over the layer of water
What are esters ?
How does ethanoic acid react with sodium hydrogen carbonate? Give equation of the reaction which takes place.
How is ethanoic acid obtained from ethanol? Write down the chemical equation of the reaction involved.
How would you distinguish between ethanol and ethanoic acid by chemical test?
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is a constituent of vinegar?(e) Which compound is a constituent of vinegar?
Write the names of three compounds which can be oxidised directly or in stages to produce acetic acid.
Define glacial acetic acid?
Fill in the blank with appropriate word/words.
Vinegar is a solution of about ________per cent ________in water.
Write balanced euation for the following :
Ethane is burnt in air.
Distinguish between esterification and saponification reactions with the help of chemical equations for each.
Write the answers to the questions by observing the following figure.

- Write the chemical reaction shown in the figure above in the form of a balanced equation.
- Write the name of the gas coming out of the large test tube in the above chemical reaction.
- Why do small bubbles appear in the small test tube?
- What is the change in colour of lime water?
Ethanoic acid turns ______ litmus to ______
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Why is glacial acetic acid called so?
Write an equation showing saponification.
Write the chemical equation for the following:
Saponification Reaction
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Observe the diagram given below and answer the questions:

- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
