Advertisements
Advertisements
प्रश्न
Three organic compounds A, B and C have the following molecular formulae: C4H10O
Which compound contains a carboxyl group? Write its name and structural formula.
Advertisements
उत्तर
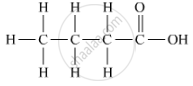
The organic compound 'A' contains a carboxyl group. The name of the acid is butanoic acid, and its formula is C3H7COOH. Its structural formula is as follows:
APPEARS IN
संबंधित प्रश्न
What are esters ?
Fill in the blanks from the choices given within brackets:
The basicity of acetic acid is-------- (3, 1, 4).
What is the common name of methanol?
What is the physical state of CH3COOH?
Describe one reaction of a carboxylic acid.
How would you distinguish between ethanol and ethanoic acid by chemical test?
Consider the following organic compounds:
HCHO, C2H5OH, C2H6, CH3COOH, C2H5CI
Choose two compounds which can react in the presence of conc. H2SO4 to form an ester. Give the name and formula of the ester formed.
An organic acid X is a liquid, which often freezes during winter time in cold countries, having the molecular formula C2H4O2. On warming it with methanol in the presence of a few drops of concentrated sulphuric acid, a compound Y with a sweet smell is formed.
(a) Identify X and Y. Also write their formulae showing the functional group present in them.
(b) Write a chemical equation for the reaction involved.
Give two tests to show that CH3COOH is acidic in nature ?
A student takes 2 mL acetic acid in a dry test tube and adds a pinch of sodium hydrogen carbonate to it. He makes the following observations:
I. A colourless and odourless gas evolves with a brisk effervescence.
II. The gas turns lime water milky when passed through it.
III. The gas burns with an explosion when a burning splinter is brought near it.
IV. The gas extinguishes the burning splinter that is brought near it.
The correct observations are:
(A) I, II, and III
(B) II, III and IV
(C) III, IV and I
(D) IV, I and II
On adding 2 mL acetic acid to 2 mL of water in a test tube, it was observed that
(A) a clear and transparent solution is formed
(B) a white precipitate is formed almost immediately
(C) two separate layers were formed
(D) a colourless and odourless gas is evolved
Write the important uses of acetic acid.
The functional group present in acetic acid is ______.
Convert ethane to acetic acid.
Write the characteristics of ethanoic acid.
Ethanoic acid turns ______ litmus to ______
Ethanoic acid is also known as which of these?
A spatula full of sodium carbonate is taken in a test tube and 2 mL of dilute ethanoic acid is added to it.
Write a chemical equation for the reaction.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
