Advertisements
Advertisements
प्रश्न
A student takes 2 mL acetic acid in a dry test tube and adds a pinch of sodium hydrogen carbonate to it. He makes the following observations:
I. A colourless and odourless gas evolves with a brisk effervescence.
II. The gas turns lime water milky when passed through it.
III. The gas burns with an explosion when a burning splinter is brought near it.
IV. The gas extinguishes the burning splinter that is brought near it.
The correct observations are:
(A) I, II, and III
(B) II, III and IV
(C) III, IV and I
(D) IV, I and II
Advertisements
उत्तर
(D)
When 2 mL acetic acid is taken in a dry test tube and a pinch of sodium hydrogen carbonate is added to it, a colourless and odourless gas evolves with a brisk effervescence which is CO2.
When CO2 is passed through lime water it turns lime water milky. 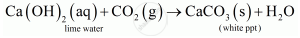
CO2 also extinguishes the burning splinter when it is brought near it.
APPEARS IN
संबंधित प्रश्न
Choose the correct alternative and rewrite the following:
Acetic acid ..................
Give the common names and IUPAC names of the following compounds of CH3COOH.
What type to compound is CH3COOH?
Name the product formed and give the appropriate chemical equation for the following:
Sodium reacting with ethyl alcohol.
Write the names of the first three members of the carboxylic acid series.
Name the functional group present in the following compound:
HCOOH
Give balanced chemical equations for the following conversion :
Ethanoic acid to ethyl ethanote
Ethanoic acid _________.
A student while observing the properties of acetic acid would report that this smells like ______.
Ester is formed by the reaction between ______.
