Advertisements
Advertisements
प्रश्न
Complete the following equation:
`CH_3 COOH + CH_2 H_5 OH` 
Advertisements
उत्तर
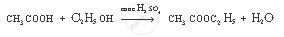
APPEARS IN
संबंधित प्रश्न
Ethanoic acid has a .................... odour.
(a) Rotten eggs
(b) Pungent
(c) Vinegar-like
(d) Mild
The molecular formula of acetic acid is _________ .
(a) CH3COOH
(b) CH3 – CH3
(c) C6H6
(d) C2H4
What do you observe when you drop a few drops of acetic acid to test tubes containing
(a) phenolphthalein
(b) distilled water
(c) universal indicator
(d) sodium hydrogen carbonate powder
Complete the following chemical equation:
\[\ce{CH3COOC2H5 + NaOH ->}\]
Complete the following chemical equations : C2H5OH + Na →
When you add a few drops of acetic acid to a test-tube containing sodium bicarbonate powder, which one of the following is your observation?
(A) No reaction takes place
(B) A colourless gas with pungent smell is released with brisk effervescence
(C) A brown coloured gas is released with brisk effervescence
(D) Formation of bubbles of a colourless and odourless gas
Give balanced chemical equations for Sodium ethanoate to methane.
Fill in the blanks from the choices given within brackets:
The basicity of acetic acid is-------- (3, 1, 4).
Give the common names and IUPAC names of the following compounds of HCOOH.
Give the name and structural formula of one homologue of HCOOH.
What is the common name of methanol?
Esters are sweet-smelling substances and are used in making perfumes. Describe an activity for the preparation of an ester with the help of a well labelled diagram. Write an equation for the chemical reaction involved in the formation of the ester. Also write the names of all the substances involved in the process of esterification.
State any two uses of esters.
Which of the following observations is true about dilute solution of acetic acid?
(A) It smells like vinegar and turns red litmus blue
(B) It smells like onion and turns blue litmus red
(C) It smells like orange and turns red litmus blue
(D) It smells like vinegar and turns blue litmus red
Acetic acid smells like:
(1) a banana
(2) vinegar
(3) an orange
(4) a lemon
Explain the following term with example.
Monomer
Explain the following term with an example.
Oxidant
A few drops of ethanoic acid were added to solid sodium carbonate. The observation made was that ______.
Reshu by mistake forgot to label the two test tubes containing Ethanol and Ethanoic acid. Suggest an experiment to identify the substances correctly? Illustrate the reactions with the help of chemical equations.
What happens when a small piece of sodium is dropped in ethanol? Write the equation for this reaction.
