Advertisements
Advertisements
प्रश्न
Complete the following equation:
`CH_3 CH_2 OH` 
Advertisements
उत्तर
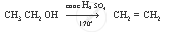
APPEARS IN
संबंधित प्रश्न
State one relevant observation for the following
When the gaseous product obtained by dehydration of ethyl alcohol is passed through bromine water.
What is the common name of methanal?
What is the molecular formula and structure of the alcohol which can be thought to be derived from pentane?
What is meant by a functional group? Explain with an example.
State two used of ethanol (other than as a fuel).
What is meant by denatured alcohol? What is the need to denature alcohol?
How would you test for an alcohol?
What happens when ethanol is oxidised with alkaline potassium permanganate (or acidified potassium dichromate)? Write the equation of the reaction involved.
When ethanol reacts with sodium metal, it forms two products. These products are:
(a) sodium ethanaoate and oxygen
(b) sodium ethanaoate and hydrogen
(c) sodium ethoxide and oxygen
(d) sodium ethoxide and hydrogen
How is the spurious alcohol obtained?
how can we prepare ethane by reduction of a halogen compound?
Name the following:
Oxidising agent used to convert ethanol to acetic acid.
The product formed when ethene gas reacts with water in the presence of sulphuric acid is ______.
Give the structural formula of the following:
Ethanoic acid
Answer the following question.
Write the chemical formula and name of the compound which is the active ingredient of all alcoholic drinks. List its two uses.
Write the chemical equation and name of the product formed when this compound reacts with -
(i) Sodium metal
(ii) hot concentrated sulphuric acid
Ethanol is soluble in water in all proportions.
100% pure ethanol is called ______.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
