Advertisements
Advertisements
Question
Complete the following equation:
`CH_3 CH_2 OH` 
Advertisements
Solution
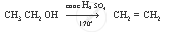
APPEARS IN
RELATED QUESTIONS
Give other two names of ethanol.
Give the structural formula of ethanol.
Give a balanced chemical equation for Action of alcoholic KOH on bromethane
The catalyst used in the contact process is
(A) Copper
(B) Iron
(C) Vanadium pentoxide
(D) Manganese dioxide
Fill in the following blank with suitable word.
The functional group present in ethanol is ...............
Give the lab. Prepared of ethyl alcohol .
Vinegar is prepared by the bacterial oxidation of ______.
how can we prepare ethane by reduction of a halogen compound?
Write the equations of chlorination of ethane
How will you bring about following conversions:
(a) Methane to methanol
(b) Ethane to ethanol (acetaldehyde)
(c) Methane to methanoic acid
Name the following:
Oxidising agent used to convert ethanol to acetic acid.
Give the structural formula of the following:
Ethanoic acid
Write the equation for the following lab preparation:
Methanol from iodomethane.
The organic compound obtained as the end product of the fermentation of a sugar solution is ______.
Write any two physical properties of Ethanol.
What are hydrocarbons? Give one example.
Write the uses of ethanol.
Write the chemical equation for the following:
Oxidation of ethanol
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
