Advertisements
Advertisements
प्रश्न
Complete the following equation:
`CH_3 CH_2 OH` 
Advertisements
उत्तर
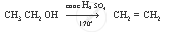
APPEARS IN
संबंधित प्रश्न
Give other two names of ethanol.
Write the name and structural formula of the compound obtained when ethanol is heated at 443 K with excess of conc. H2SO4. Also write chemical equation for the reaction stating the role of conc. H2SO4 in it.
Give a balanced chemical equation for Preparation of ethane from Sodium propionate
Write a balanced chemical equation for Burning of ethane in the plentiful supply of air.
Give the structural formula of Ethanol
Fill in the following blank with suitable word.
The functional group present in ethanol is ...............
Name one liquid carbon compound which is being used as an additive in petrol in some countries.
What would be observed on adding a 5% alkaline potassium permanganate solution drop by drop to some warm ethanol in a test-tube? Write the name of the compound formed during the chemical reaction. Also write chemical equation of the reaction which takes place.
Identify the functional group of the CH3COOH ?
What is meant by a functional group in carbon compounds?
The product formed when ethene gas reacts with water in the presence of sulphuric acid is ______.
Give the structural formula of the following:
Ethanoic acid
State how the following conversion can be carried out:
Ethene to ethyl alcohol
An organic compound ‘A’ is widely used as a preservative and has the molecular formula C2H4O2. This compound reacts with ethanol to form a sweet-smelling compound ‘B’.
Write the chemical equation for its reaction with ethanol to form compound ‘B’.
Ethanol reacts with sodium and forms two products. These are
Draw the electron dot structure of ethyne and also draw its structural formula
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
