Advertisements
Advertisements
प्रश्न
Complete the following equation:
`CH_3 COOH + CH_2 H_5 OH` 
Advertisements
उत्तर
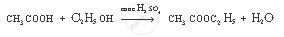
APPEARS IN
संबंधित प्रश्न
Complete the following chemical equations: C2H5OH+O2→
Choose the correct alternative and rewrite the following:
Acetic acid ..................
Give a reason for Conductivity of dilute hydrochloric acid is greater than that of acetic acid
Write the IUPAC names, common names and formulae of the first two members of the homologous series of carboxylic acids.
What type to compound is CH3COOH?
Name two oxidising agents which can oxidise ethanol to ethanoic acid.
How does ethanoic acid react with sodium hydrogen carbonate? Give equation of the reaction which takes place.
Name the compound formed when acetic acid and ethanol react together.
Acetic acid smells like:
(1) a banana
(2) vinegar
(3) an orange
(4) a lemon
How is ethyne prepared in the laboratory?
Fill in the blank with appropriate word/words.
Denatured alcohol is a mixture of _____ and _______
When acetaldehyde is oxidized with acidified potassium dichromate, it forms ______.
Identify the term or substance based on the descriptions given below:
Ice like crystals formed on cooling an organic acid sufficiently.
Draw the structure formula of ethyne.
Ethanoic acid _________.
A student while observing the properties of acetic acid would report that this smells like ______.
Bubbles are seen in the test tube during the preparation of lime water.
Why is glacial acetic acid called so?
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
