Advertisements
Advertisements
प्रश्न
Draw the electron dot structure of ethyne and also draw its structural formula
Advertisements
उत्तर
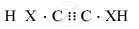
\[\ce{H - C ≡ C - H}\]
APPEARS IN
संबंधित प्रश्न
Identify the term/substance in the following:
The catalyst used in the conversion of ethyne to ethane.
Write a balanced chemical equation for Burning of ethane in the plentiful supply of air.
The catalyst used in the contact process is
(A) Copper
(B) Iron
(C) Vanadium pentoxide
(D) Manganese dioxide
The usual disease caused by the excessive drinking of alcohol over a long period of time is:
(a) diabetes
(b) cataract
(c) cirrhosis
(d) arthritis
Identify the functional group of the HCHO ?
Write the name and molecular formula of an organic compound having its name suffixed with 'ol' and having two carbon atoms in its molecule. Write balanced chemical equation to indicate what happens when this compound is heated with excess conc. H2SO4 and the name of main product formed. Also state the role of conc. H2SO4 in the reaction.
Name the following:
100% ethanol
Give three uses of ethanol
Write a balanced chemical equation for the following:
Ethanol reacts with sodium at room temperature.
From the given list, name the compound with −OH as part of its structure.
The organic compound obtained as the end product of the fermentation of a sugar solution is ______.
State how the following conversion can be carried out:
Ethyl alcohol to ethene.
Write any two physical properties of Ethanol.
Which of the following are used as anesthetics?
What is the role of concentrated Sulphuric acid when it is heated with Ethanol at 443 K. Give the reaction involved.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
