Advertisements
Advertisements
Question
Draw the electron dot structure of ethyne and also draw its structural formula
Advertisements
Solution
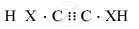
\[\ce{H - C ≡ C - H}\]
APPEARS IN
RELATED QUESTIONS
Write a balanced chemical equations for Heating of Ethanol at 170°C in the presence of conc. Sulphuric acid
Give the structural formula of Ethanol
The catalyst used in the contact process is
(A) Copper
(B) Iron
(C) Vanadium pentoxide
(D) Manganese dioxide
Three organic compounds A, B and C have the following molecular formulae: C4H8O2
Which compound contains an alcohol group? Write its name and structural formula.
Give the harmful effects of drinking alcohol.
What would be observed on adding a 5% alkaline potassium permanganate solution drop by drop to some warm ethanol in a test-tube? Write the name of the compound formed during the chemical reaction. Also write chemical equation of the reaction which takes place.
How is the absolute alcohol obtained?
Write a chemical reaction to show the dehydration of ethanol.
How does ethanol reacts with
(a) Metallic sodium
(b) Acetic acid
(c) Conc.H2S04
Ethanol can be converted to ethene which can be changed to ethane.
Choose the correct word or phrase from the options given below to complete the following sentences:
Converting ethanol to ethene requires the use of_________(Concentrated hydrochloric acid, concentrated nitric cid, concentrated sulphuric acid).
The product formed when ethene gas reacts with water in the presence of sulphuric acid is ______.
Write a balanced chemical equation for the following:
Ethanol reacts with sodium at room temperature.
Write the equation for the following lab preparation:
Methanol from iodomethane.
Why is the conversion of ethanol to ethanoic acid considered an oxidation reaction? Write the oxidising agent used in the reaction involved.
Write the uses of ethanol.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
