Advertisements
Advertisements
प्रश्न
Draw the electron dot structure of ethyne and also draw its structural formula
Advertisements
उत्तर
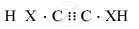
\[\ce{H - C ≡ C - H}\]
APPEARS IN
संबंधित प्रश्न
Give the structural formula of ethanol.
Write the molecular formula of the 2nd and the 3rd member of the homologous series whose first member is methane.
Identify the term/substance in the following:
The catalyst used in the conversion of ethyne to ethane.
Fill in the following blank with suitable word.
The functional group present in ethanol is ...............
Write the name of the following functional group:

Draw the structures for the following compounds of Pentanal
Describe one reaction of ethanol.
Complete the following equation:
`CH_3 CH_2 OH` 
What happens when ethanol reacts with ethanoic acid in the presence of a little of concentrated sulphuric acid? Write equation of the reaction involved.
Which of the following can damage optic nerve leading to blindness, if taken internally?
(a) CH3COOH
(b) C2H5OH
(c) NaHCO3
(d) CH3OH
How is acetic acid prepared from ethanol?
Write a balanced chemical equation for the following:
Ethanol under high pressure and low temperature is treated with acidified potassium dichoromate.
The organic compound obtained as the end product of the fermentation of a sugar solution is ______.
Why is the conversion of ethanol to ethanoic acid considered an oxidation reaction? Write the oxidising agent used in the reaction involved.
Complete the following activity.
| Boiling point of ethanol | → | |
| General name of ethanol | → | |
| Use of ethanol | → | |
| Boiling point of ethanoic acid | → | |
| Melting point of pure ethanoic acid | → |
How is ethanol manufactured from sugarcane?
Ethene is formed when ethanol at 443 K is heated with excess of concentrated sulphuric acid. What is the role of sulphuric acid in this reaction? Write the balanced chemical equation of this reaction.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the reaction:
Combustion of ethanol.
