Advertisements
Advertisements
प्रश्न
Draw the structures for the following compounds of Pentanal
Advertisements
उत्तर
Molecular structures of the compounds are as follows:
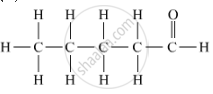
APPEARS IN
संबंधित प्रश्न
Why is ethanol used as a fuel?
How would you convert of thanol into ethene?
What would be observed on adding a 5% alkaline potassium permanganate solution drop by drop to some warm ethanol in a test-tube? Write the name of the compound formed during the chemical reaction. Also write chemical equation of the reaction which takes place.
State the method of preparation of ethanol by hydrolysis of ethene.
Identify the functional group of the HCHO ?
How is ethane prepared by Wurtz reaction?
Give the necessary conditions and equations of getting ethanol from:
(a) Alkyl halide
(b) An ethene
How does ethanol reacts with
(a) Metallic sodium
(b) Acetic acid
(c) Conc.H2S04
Addition reactions and substitution reactions are types of organic reactions.Which type of reaction is shown by?
Ethene?
The product formed when ethene gas reacts with water in the presence of sulphuric acid is ______.
The organic compound obtained as the end product of the fermentation of a sugar solution is ______.
Draw the structures for ethanol.
Write the characteristics of ethanol.
100% pure ethanol is called ______.
How is ethanol manufactured from sugarcane?
Give the balanced chemical equation of the following reaction:
Combustion of ethanol
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
