Advertisements
Advertisements
प्रश्न
What happens when ethanol reacts with ethanoic acid in the presence of a little of concentrated sulphuric acid? Write equation of the reaction involved.
Advertisements
उत्तर
When ethanol reacts with ethanoic acid in the presence of a few drops of concentrated sulphuric acid, a sweet smelling ester named ethyl ethanoate is formed.
The chemical equation for the above reaction is:
`CH_3 COOH+_2 H_5 OH` 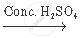 `CH_3 COOC_2 H_5 + H_2 O`
`CH_3 COOC_2 H_5 + H_2 O`
APPEARS IN
संबंधित प्रश्न
Give the structural formula of ethanol.
Identify the term/substance in the following:
The catalyst used in the conversion of ethyne to ethane.
Give a balanced equation for the preparation of ethanol from monochloroethane and aq. sodium hydroxide.
Fill in the following blank with suitable word:
The next homologue of C2H5OH is ...............
Organic compounds having functional group are known as
functional group are known as
Write the name of the following functional group:

Describe one reaction of ethanol.
State two used of ethanol (other than as a fuel).
What is meant by denatured alcohol? What is the need to denature alcohol?
State the method of preparation of ethanol by hydrolysis of ethene.
How is acetic acid prepared from ethanol?
Name the oxidising agent used for the conversion of ethanol to ethanoic acid. Distinguish between ethanol and ethanoic acid on the basis of (i) litmus test, (ii) reaction with sodium carbonate
In a tabular form, differentiate between ethanol and ethanoic acid under the following heads:
(i) Physical state
(ii) Taste
(iii) NaHCO3 test
(iv) Ester test
Write a balanced chemical equation for the following:
Ethanol under high pressure and low temperature is treated with acidified potassium dichoromate.
The organic compound obtained as the end product of the fermentation of a sugar solution is ______.
Sodium is stored under kerosene. Give reason.
Why is the conversion of ethanol to ethanoic acid considered an oxidation reaction? Write the oxidising agent used in the reaction involved.
How is ethene prepared from ethanol? Give the reaction involved in it.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
