Advertisements
Advertisements
Question
What happens when ethanol reacts with ethanoic acid in the presence of a little of concentrated sulphuric acid? Write equation of the reaction involved.
Advertisements
Solution
When ethanol reacts with ethanoic acid in the presence of a few drops of concentrated sulphuric acid, a sweet smelling ester named ethyl ethanoate is formed.
The chemical equation for the above reaction is:
`CH_3 COOH+_2 H_5 OH` 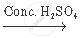 `CH_3 COOC_2 H_5 + H_2 O`
`CH_3 COOC_2 H_5 + H_2 O`
APPEARS IN
RELATED QUESTIONS
Give the structural formula of ethanol.
Give a balanced chemical equation for Preparation of ethane from Sodium propionate
Identify the term/substance in the following:
The catalyst used in the conversion of ethyne to ethane.
State one relevant observation for the following
When the gaseous product obtained by dehydration of ethyl alcohol is passed through bromine water.
Fill in the blanks from the choices given within brackets:
The compound formed when ethanol reacts with sodium is____ (sodium ethanoate, sodium ethoxide, sodium propanoate).
When ethanol is heated with concentrated sulphuric acid at 170°C, it gets converted into ethene. In this reaction, concentrated sulphuric acid acts as:
(a) oxidising agent
(b) catalyst
(c) dehydrating agent
(d) reducing agent
One of the following substances is not added to make denatured alcohol. This is:
(a) methyl alcohol
(b) copper sulphate
(c) chloroform
(d) pyridine
How is ethanol prepared by fermentation?
Give the lab. Prepared of ethyl alcohol .
How do the boiling point and melting point change in the homologous series of alcohols?
How is the spurious alcohol obtained?
An organic compound 'P' is a constituent of wine. 'P' on reacting with acidified K2Cr2O7 forms another compound 'Q'. When a piece of sodium is added to 'Q' a gas 'R' evolves which burns with a pop sound. Identify P, Q and R and write the chemical equations of the reactions involved.
How does ethanol reacts with
(a) Metallic sodium
(b) Acetic acid
(c) Conc.H2S04
Name an organic compound which is dehydrated to produce ethene.
What are hydrocarbons? Give one example.
Colorless ethanol is in liquid state at room temperature.
Which of the following are used as anesthetics?
The organic compounds having functional group are known as ______.
How is ethene prepared from ethanol? Give the reaction involved in it.
