Advertisements
Advertisements
प्रश्न
What happens when ethanol reacts with ethanoic acid in the presence of a little of concentrated sulphuric acid? Write equation of the reaction involved.
Advertisements
उत्तर
When ethanol reacts with ethanoic acid in the presence of a few drops of concentrated sulphuric acid, a sweet smelling ester named ethyl ethanoate is formed.
The chemical equation for the above reaction is:
`CH_3 COOH+_2 H_5 OH` 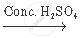 `CH_3 COOC_2 H_5 + H_2 O`
`CH_3 COOC_2 H_5 + H_2 O`
APPEARS IN
संबंधित प्रश्न
............ is a functional group of the alcohol.
The catalyst used in the contact process is
(A) Copper
(B) Iron
(C) Vanadium pentoxide
(D) Manganese dioxide
Name the hydrocarbon formed when ethanol is heated with conc. H2SO4 at 170°C? What is this reaction known as?
State two used of ethanol (other than as a fuel).
What would be observed on adding a 5% alkaline potassium permanganate solution drop by drop to some warm ethanol in a test-tube? Write the name of the compound formed during the chemical reaction. Also write chemical equation of the reaction which takes place.
When ethanol is heated with concentrated sulphuric acid at 170°C, it gets converted into ethene. In this reaction, concentrated sulphuric acid acts as:
(a) oxidising agent
(b) catalyst
(c) dehydrating agent
(d) reducing agent
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is used to sterilise wounds and syringes?
Write a chemical reaction to show the dehydration of ethanol.
Name a solid which can be used, instead of conc H2SO4 to prepare ethylene by dehydration of ethanol.
Write any two physical properties of Ethanol.
Draw the structures for ethanol.
3 mL of ethanol is taken in a test tube and warmed gently in a water bath. A 5% solution of alkaline potassium permanganate is added to the first drop by drop to this solution, then in excess.
- How is a 5% solution of KMnO4 prepared?
- State the role of alkaline potassium permanganate in this reaction. What happens on adding it in excess?
- Write the chemical equation of this reaction.
Write the molecular formula of the given compound.
Ethanol
A gas is evolved when ethanol reacts with sodium. Name the gas evolved and also write the balanced chemical equation of the reaction involved.
How would you bring about the following conversions? Name the process and write the reaction involved.
Name the compound formed when ethanol is heated at 443 K in the presence of conc. \[\ce{H2SO4}\] and draw its electron dot structure.
State the role of conc. \[\ce{H2SO4}\] in the reaction.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
