Advertisements
Advertisements
प्रश्न
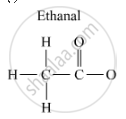
Draw the structures for the following compounds:
Ethanal
Advertisements
उत्तर
Molecular structures of the compounds are as follows:[
APPEARS IN
संबंधित प्रश्न
Give two properties of ethanol.
Write the molecular formula of the 2nd and the 3rd member of the homologous series whose first member is methane.
The compound formed when ethene reacts with Hydrogen is ________. (CH4, C2H6, C3H8)
Write balanced chemical equations for Preparation of ethanol from Ethyl Chloride
Draw the structure for the following compound:
Butanal
Why is ethanol used as a fuel?
Explain why, methanol is much more dangerous to drink than ethanol.
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is used to sterilise wounds and syringes?
How is ethanol prepared by fermentation?
Identify the functional group of the CH3OH ?
List in tabular form three physical and two chemical properties on the basis of which ethanol and ethanoic acid can be differentiated.
Ethanol can be converted to ethene which can be changed to ethane.
Choose the correct word or phrase from the options given below to complete the following sentences:
Converting ethanol to ethene requires the use of_________(Concentrated hydrochloric acid, concentrated nitric cid, concentrated sulphuric acid).
Write a balanced chemical equation for the following:
Ethanol under high pressure and low temperature is treated with acidified potassium dichoromate.
The organic compound obtained as the end product of the fermentation of a sugar solution is ______.
Sodium is stored under kerosene. Give reason.
Why is the conversion of ethanol to ethanoic acid considered an oxidation reaction? Write the oxidising agent used in the reaction involved.
C2H5OH + 3O2 → 2CO2 + 3H2O is a ______
How is ethanoic acid prepared from ethanol? Give the chemical equation.
______ react with dil. HNO3 to evolve hydrogen gas.
Draw electron dot structure of Ethanol.
