Advertisements
Advertisements
Question
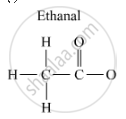
Draw the structures for the following compounds:
Ethanal
Advertisements
Solution
Molecular structures of the compounds are as follows:[
APPEARS IN
RELATED QUESTIONS
Match the formulae in group A with appropriate names from group B:
Group A: CH3COOH, CH3CHO, CH3OH
Group B: Ethanol, Methanol, Ethanal, Ethanoic acid
Describe one reaction of ethanol.
Write the chemical equation of the reaction which takes place during the burning of ethanol in air.
What happens when ethanol reacts with ethanoic acid in the presence of a little of concentrated sulphuric acid? Write equation of the reaction involved.
When ethanol is heated with concentrated sulphuric acid at 170°C, it gets converted into ethene. In this reaction, concentrated sulphuric acid acts as:
(a) oxidising agent
(b) catalyst
(c) dehydrating agent
(d) reducing agent
How is ethanol prepared by fermentation?
Give the lab. Prepared of methyl alcohol.
Name the product formed when ethanol reacts with acetic acid. Give an equation. What is the name given to this type of reaction?
What is meant by a functional group in carbon compounds?
In a tabular form, differentiate between ethanol and ethanoic acid under the following heads:
(i) Physical state
(ii) Taste
(iii) NaHCO3 test
(iv) Ester test
how can we prepare ethane by reduction of a halogen compound?
Give three uses of ethanol
The product formed when ethene gas reacts with water in the presence of sulphuric acid is ______.
Write a balanced chemical equation for the following:
Ethanol under high pressure and low temperature is treated with acidified potassium dichoromate.
Write a balanced chemical equation for the following:
Ethanol reacts with sodium at room temperature.
What are hydrocarbons? Give one example.
C2H5OH + 3O2 → 2CO2 + 3H2O is a ______
How is ethene prepared from ethanol? Give the reaction involved in it.
Give the balanced chemical equation of the reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
