Advertisements
Advertisements
प्रश्न
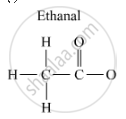
Draw the structures for the following compounds:
Ethanal
Advertisements
उत्तर
Molecular structures of the compounds are as follows:[
APPEARS IN
संबंधित प्रश्न
Give two properties of ethanol.
A carbon compound 'P' on heating with excess conc. H2SO4 forms another carbon compound 'Q' which on addition of hydrogen in the presence of nickel catalyst forms a saturated carbon compound 'R'. One molecule of 'R' on combustion forms two molecules of carbon dioxide and three molecules of water. Identify P, Q and R and write chemical equations for the reactions involved
Fill in the blanks from the choices given within brackets:
The compound formed when ethanol reacts with sodium is____ (sodium ethanoate, sodium ethoxide, sodium propanoate).
Give the structural formula of Ethanol
Give one exampleof the compound having the following functional group:
Alcohol group
Draw the structures for the following compounds:
Propanal
Complete the following equation:
`CH_3 CH_2 OH` 
Why is ethanol used as a fuel?
Explain why, methanol is much more dangerous to drink than ethanol.
Name the oxidising agent used for the conversion of ethanol to ethanoic acid. Distinguish between ethanol and ethanoic acid on the basis of (i) litmus test, (ii) reaction with sodium carbonate
How will you bring about following conversions:
(a) Methane to methanol
(b) Ethane to ethanol (acetaldehyde)
(c) Methane to methanoic acid
Write the balanced equations to convert:
(a) Ethanol to ethene
(b) Ethene to ethanol
Addition reactions and substitution reactions are types of organic reactions.Which type of reaction is shown by?
Ethene?
Write a balanced chemical equation for the following:
Ethanol under high pressure and low temperature is treated with acidified potassium dichoromate.
Choose the correct alternative and rewrite the following:
To observe the hydro and clearly, Raju should see it first under the low power lens and then under the high power lens in order to see _____________
Ethene is formed when ethanol at 443 K is heated with excess of concentrated sulphuric acid. What is the role of sulphuric acid in this reaction? Write the balanced chemical equation of this reaction.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
