Advertisements
Advertisements
प्रश्न
Draw the structures for the following compounds of Pentanal
Advertisements
उत्तर
Molecular structures of the compounds are as follows:
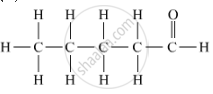
APPEARS IN
संबंधित प्रश्न
Write the name of the following functional group:

Complete the following equation:
`CH_3 CH_2 OH` 
When ethanol reacts with sodium metal, it forms two products. These products are:
(a) sodium ethanaoate and oxygen
(b) sodium ethanaoate and hydrogen
(c) sodium ethoxide and oxygen
(d) sodium ethoxide and hydrogen
How is ethanol prepared by fermentation?
What would you see, when ethyne is bubbled through a solution of bromine in carbon tetrachloride?
Identify the functional group of the CH3OH ?
An organic compound 'P' is a constituent of wine. 'P' on reacting with acidified K2Cr2O7 forms another compound 'Q'. When a piece of sodium is added to 'Q' a gas 'R' evolves which burns with a pop sound. Identify P, Q and R and write the chemical equations of the reactions involved.
In a tabular form, differentiate between ethanol and ethanoic acid under the following heads:
(i) Physical state
(ii) Taste
(iii) NaHCO3 test
(iv) Ester test
Name the following:
Oxidising agent used to convert ethanol to acetic acid.
Ethanol can be converted to ethene which can be changed to ethane.
Choose the correct word or phrase from the options given below to complete the following sentences:
The conversion of ethanol to ehane is an example of_________(dehydration, dehydrogenation)
Write balanced equations to show the preparation of the following:
Ethene from ethanol
From the given list, name the compound with −OH as part of its structure.
Sodium is stored under kerosene. Give reason.
Write the molecular formula of the given compound.
Sodium ethoxide
Write the molecular formula of the given compound.
Ethanol
How is ethene prepared from ethanol? Give the reaction involved in it.
Name the compound formed when ethanol is heated at 443 K in the presence of conc. \[\ce{H2SO4}\] and draw its electron dot structure.
State the role of conc. \[\ce{H2SO4}\] in the reaction.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
