Advertisements
Advertisements
प्रश्न
Give balanced chemical equations for Sodium ethanoate to methane.
Advertisements
उत्तर
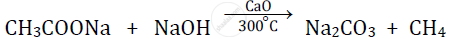
APPEARS IN
संबंधित प्रश्न
A student adds 2 mL of acetic acid to a test tube containing 2 mL of distilled water. He then shakes the test tube well and leaves it to settle for some time. After about 5 minutes he observes that in the test tube there is :
(A) a clear transparent colourless solution
(B) a clear transparent pink solution
(C) a precipitate settling at the bottom of the test tube
(D) a layer of water the layer of acetic acid
Complete the following equation:
`CH_3 COOH + CH_2 H_5 OH` 
What do you notice when acetic acid reacts with metals?
Why is pure acetic acid also known as glacial acetic acid?
Explain the following reaction with an example.
Esterification
Ethanoic acid is also known as which of these?
When ethanoic acid is treated with NaHCO^ the gas evolved is ______.
What happens when a small piece of sodium is dropped in ethanol? Write the equation for this reaction.
Write the chemical equation for the ethanol to ethanoic acid of an oxidation reaction.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
