Advertisements
Advertisements
Question
Give balanced chemical equations for Sodium ethanoate to methane.
Advertisements
Solution
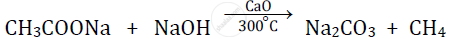
APPEARS IN
RELATED QUESTIONS
Write three different chemical reactions showing the conversion of ethanoic acid to sodium ethanoate. Write balanced chemical equation in each case. Write the name of the reactants and the products other ethanoic acid and sodium ethanoate in each case.
Draw the structure for the following compound.
Ethanoic acid
What type of compound is formed when a carboxylic acid reacts with an alcohol in the presence of conc. H2SO4?
Name the product formed and give the appropriate chemical equation for the following:
Sodium reacting with ethyl alcohol.
What is the action of acetic acid with litmus paper?
How Will You Carry Out the Following Conversions?
Ehane to acitic acid
With a labelled diagram describe in brief an activity to show the formation of ester.
Ethanoic acid is also known as which of these?
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
