Advertisements
Advertisements
प्रश्न
Give balanced chemical equations for Sodium ethanoate to methane.
Advertisements
उत्तर
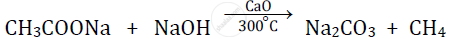
APPEARS IN
संबंधित प्रश्न
Ethanoic acid has a .................... odour.
(a) Rotten eggs
(b) Pungent
(c) Vinegar-like
(d) Mild
For preparing soap in the laboratory we require an oil and a base. Which of the following combinations of an oil and a base would be best suited for the preparation of soap?
(a) Castor oil and calcium hydroxide
(b) Turpentine oil and sodium hydroxide
(c) Castor oil and sodium hydroxide
(d) Mustard oil and calcium hydroxide
Name the following:
The distinctive reaction that takes place when ethanol is treated with acetic acid.
Give the structural formulae of acetic acid.
Name the compound formed when acetic acid and ethanol react together.
Write the molecular formula of the given compound.
Ethanoic acid
Give the balanced chemical equation of the following reaction:
Evolution of carbon dioxide by the action of ethanoic acid with NaHCO3.
Ethanoic acid is also known as which of these?
Which of these is not an organic acid?
Bubbles are seen in the test tube during the preparation of lime water.
