Advertisements
Advertisements
Question
A student takes 2 mL acetic acid in a dry test tube and adds a pinch of sodium hydrogen carbonate to it. He makes the following observations:
I. A colourless and odourless gas evolves with a brisk effervescence.
II. The gas turns lime water milky when passed through it.
III. The gas burns with an explosion when a burning splinter is brought near it.
IV. The gas extinguishes the burning splinter that is brought near it.
The correct observations are:
(A) I, II, and III
(B) II, III and IV
(C) III, IV and I
(D) IV, I and II
Advertisements
Solution
(D)
When 2 mL acetic acid is taken in a dry test tube and a pinch of sodium hydrogen carbonate is added to it, a colourless and odourless gas evolves with a brisk effervescence which is CO2.
When CO2 is passed through lime water it turns lime water milky. 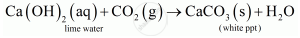
CO2 also extinguishes the burning splinter when it is brought near it.
APPEARS IN
RELATED QUESTIONS
Complete the following equation:
`CH_3 COOH + CH_2 H_5 OH` 
How would you distinguish between ethanol and ethanoic acid by chemical test?
What do you observe when acetic acid is added to sodium bicarbonate?
Write the important uses of acetic acid.
Draw the structural formula of a compound with two carbon atoms in the following case:
An alkane with a carbon to carbon single bond.
Write the molecular formula of the given compound.
Ethanoic acid
Bubbles are seen in the test tube during the preparation of lime water.
Anita added a drop each of diluted acetic acid and diluted hydrochloric acid on pH paper and compared the colors. Which of the following is the correct conclusion?
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
