Advertisements
Advertisements
प्रश्न
A student takes 2 mL acetic acid in a dry test tube and adds a pinch of sodium hydrogen carbonate to it. He makes the following observations:
I. A colourless and odourless gas evolves with a brisk effervescence.
II. The gas turns lime water milky when passed through it.
III. The gas burns with an explosion when a burning splinter is brought near it.
IV. The gas extinguishes the burning splinter that is brought near it.
The correct observations are:
(A) I, II, and III
(B) II, III and IV
(C) III, IV and I
(D) IV, I and II
Advertisements
उत्तर
(D)
When 2 mL acetic acid is taken in a dry test tube and a pinch of sodium hydrogen carbonate is added to it, a colourless and odourless gas evolves with a brisk effervescence which is CO2.
When CO2 is passed through lime water it turns lime water milky. 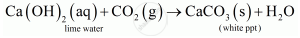
CO2 also extinguishes the burning splinter when it is brought near it.
APPEARS IN
संबंधित प्रश्न
A student adds a few drops of ethanoic acid to test tubes X, Y and Z containing aqueous solutions of sodium chloride, sodium hydroxide and sodium carbonate, respectively. If he now brings a burning splinter near the mouth of the test tubes immediately after adding ethanoic acid in each one of them, in which of the test tube or test tubes the flame will be extinguished?
(A) X and Y
(B) Y and Z
(C) X and Z
(D) only Z
Complete the following chemical equations: C2H5OH+O2→
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is most likely to be sweet-smelling?
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is added to alcohol to denature it?
The next higher homologue of ethanoic acid is ______.
A student adds 4 mL of acetic acid to a test tube containing 4 mL of distilled water. He then shakes the test tube and leaves it to settle. After about 10 minutes he observes:
(A) a layer of water over the layer of acetic acid
(B) a layer of acetic acid over the layer of water
(C) a precipitate settling at the bottom of the test tube
(D) a clear colourless solution
How is ethyne prepared in the laboratory?
How Will You Carry Out the Following Conversions?
Ehane to acitic acid
Bubbles are seen in the test tube during the preparation of lime water.
Raina while doing certain reactions observed that heating of substance ‘X’ with a vinegar-like smell with a substance ‘Y’ (which is used as an industrial solvent.) in the presence of conc. Sulphuric acid in a water bath gives a sweet-smelling liquid ‘Z’ having molecular formula C4H8O2. When heated with caustic soda (NaOH), ‘Z’ gives back the sodium salt and the compound ‘Y’.
Identify ‘X’, ‘Y’, and ‘Z’. Illustrate the changes with the help of suitable chemical equations.
