Advertisements
Advertisements
प्रश्न
An organic acid X is a liquid, which often freezes during winter time in cold countries, having the molecular formula C2H4O2. On warming it with methanol in the presence of a few drops of concentrated sulphuric acid, a compound Y with a sweet smell is formed.
(a) Identify X and Y. Also write their formulae showing the functional group present in them.
(b) Write a chemical equation for the reaction involved.
Advertisements
उत्तर
(a) The organic acid, X, which often freezes in the winters in cold countries, is ethanoic acid. It has the molecular formula CH3COOH.
The formula with its functional group is represented as:
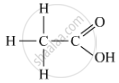
When ethanoic acid is warmed with methanol in the presence of a few drops of concentrated sulphuric acid, the compound 'Y' with sweet fragrance is formed. This compound is called methyl ethanoate, and its molecular formula is CH3COOCH3.
The formula with its functional group is represented as:
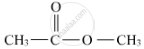
(b) The chemical equation of the above stated reaction is as follows:
`CH_3 COOH + C_2 H_5 OH` 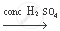 `CH_3 COOC_2 H_5 + H_2O`
`CH_3 COOC_2 H_5 + H_2O`
APPEARS IN
संबंधित प्रश्न
For preparing soap in the laboratory we require an oil and a base. Which of the following combinations of an oil and a base would be best suited for the preparation of soap?
(a) Castor oil and calcium hydroxide
(b) Turpentine oil and sodium hydroxide
(c) Castor oil and sodium hydroxide
(d) Mustard oil and calcium hydroxide
When you add about 2 ml of acetic acid to a test tube containing an equal amount of distilled water and leave the test tube to settle after shaking its contents, what will you observe in the test tube after about 5 minutes?
(A) A white precipitate settling at its bottom
(B) A clear colourless solution
(C) A layer of water over the layer of acetic acid
(D) A layer of acetic acid over the layer of water
When zinc powder is added to acetic acid ______________
(a) the mixture becomes warm
(b) a gas is evolved
(c) the colour of the mixture becomes yellow
(d) a solid settles at the bottom
Write the name and chemical formula of the simplest organic acid.
Give the name and structural formula of one homologue of HCOOH.
Write the formulae of methanoic acid.
What type of compound is formed when a carboxylic acid reacts with an alcohol in the presence of conc. H2SO4?
Choose those compounds from the following which can turn blue litmus solution red:
HCHO, CH3COOH, CH3OH, C2H5OH, HCOOH, CH3CHO
Give reasons for your choice.
Give any two uses of ethanoic acid.
If you are asked to report your observations about the following two properties of acetic acid, what would you report?
(i) Odour
(ii) Effect on litmus
A student puts a drop of acetic acid first on a blue litmus paper and then on a red litmus paper. He would observe that
(A) the red litmus paper turns colourless and there is no change in the blue litmus paper.
(B) the red litmus paper turns blue and the blue litmus paper turns red.
(C) there is no change in the red litmus paper and the blue litmus paper turns red.
(D) there is no change in the blue litmus paper and the red litmus paper turns blue.
While studying saponification reactions, the following comments were noted down by the students :
(I) Soap is a salt of fatty acids.
(II) The reaction mixture is basic in nature.
(III) In this reaction heat is absorbed.
(IV) This reaction is not a neutralisation reaction.
Which of these are the correct comments ?
(A) I and III only
(B) I, II and III
(C) II, III and IV
(D) I and II only
How will you carry out the following conversions?
Ethene to acetic acid
What type of compound is formed by the reaction between acetic acid and an alcohol?
Give balanced chemical equations for the following conversion :
Ethanoic acid to ethyl ethanote
When sodium hydrogen carbonate is added to ethanoic acid, a gas evolves. Consider the following statements about the gas evolved.
- It turns lime water milky.
- It is evolved with brisk effervescence.
- It has a smell of burning sulfur.
- It is also a by-product of respiration.
Write the chemical equation for the following:
Saponification Reaction
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Observe the diagram given below and answer the questions:

- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
