Advertisements
Advertisements
प्रश्न
An organic acid X is a liquid, which often freezes during winter time in cold countries, having the molecular formula C2H4O2. On warming it with methanol in the presence of a few drops of concentrated sulphuric acid, a compound Y with a sweet smell is formed.
(a) Identify X and Y. Also write their formulae showing the functional group present in them.
(b) Write a chemical equation for the reaction involved.
Advertisements
उत्तर
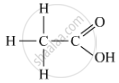
(a) The organic acid, X, which often freezes in the winters in cold countries, is ethanoic acid. It has the molecular formula CH3COOH.
The formula with its functional group is represented as:
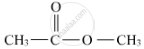
When ethanoic acid is warmed with methanol in the presence of a few drops of concentrated sulphuric acid, the compound 'Y' with sweet fragrance is formed. This compound is called methyl ethanoate, and its molecular formula is CH3COOCH3.
The formula with its functional group is represented as:
(b) The chemical equation of the above stated reaction is as follows:
`CH_3 COOH + C_2 H_5 OH`  `CH_3 COOC_2 H_5 + H_2O`
`CH_3 COOC_2 H_5 + H_2O`
APPEARS IN
संबंधित प्रश्न
Complete the following chemical equations: C2H5OH+O2→
When zinc powder is added to acetic acid ______________
(a) the mixture becomes warm
(b) a gas is evolved
(c) the colour of the mixture becomes yellow
(d) a solid settles at the bottom
Write the IUPAC names, common names and formulae of the first two members of the homologous series of carboxylic acids.
Esters are sweet-smelling substances and are used in making perfumes. Describe an activity for the preparation of an ester with the help of a well labelled diagram. Write an equation for the chemical reaction involved in the formation of the ester. Also write the names of all the substances involved in the process of esterification.
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is added to alcohol to denature it?
Ethanol can be oxidized to ethanoic acid. Write the equation and name the oxidizing agent.
If you take a pinch of sodium hydrogen carbonate powder in a test-tube and add drop-by-drop acetic acid to it, what would you observe immediately? List any two main observations.
While studying saponification reactions, the following comments were noted down by the students :
(I) Soap is a salt of fatty acids.
(II) The reaction mixture is basic in nature.
(III) In this reaction heat is absorbed.
(IV) This reaction is not a neutralisation reaction.
Which of these are the correct comments ?
(A) I and III only
(B) I, II and III
(C) II, III and IV
(D) I and II only
On adding acetic acid to sodium hydrogen carbonate in a test tube, a student observes
(A) no reaction
(B) a colourless gas with pungent smell
(C) bubbles of a colourless and odourless gas
(D) a strong smell of vinegar
On adding NaHCO3 to acetic acid, a gas is evolved which turns lime water milky due to the formation of:
(1) Calcium bicarbonate
(2) Calcium hydroxide
(3) Calcium carbonate
(4) Calcium acetate
When acetaldehyde is oxidized with acidified potassium dichromate, it forms ______.
Write a balanced chemical equation for the following:
Monochloroethane is hydrolysed with aqueous KOH.
Ethanoic acid _________.
Rewrite the following statement by selecting the correct option.
The formula of ethanoic acid is _____________.
Assertion (A): Esterification is a process in which a sweet-smelling substance is produced.
Reason (R): When esters react with sodium hydroxide an alcohol and sodium salt of carboxylic acid are obtained.
What are catalysts?
Ethanoic acid reacts with the basic salt, sodium carbonate, to form a salt, named ______, water and carbon dioxide gas.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
