Advertisements
Advertisements
Question
An organic acid X is a liquid, which often freezes during winter time in cold countries, having the molecular formula C2H4O2. On warming it with methanol in the presence of a few drops of concentrated sulphuric acid, a compound Y with a sweet smell is formed.
(a) Identify X and Y. Also write their formulae showing the functional group present in them.
(b) Write a chemical equation for the reaction involved.
Advertisements
Solution
(a) The organic acid, X, which often freezes in the winters in cold countries, is ethanoic acid. It has the molecular formula CH3COOH.
The formula with its functional group is represented as:
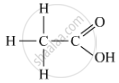
When ethanoic acid is warmed with methanol in the presence of a few drops of concentrated sulphuric acid, the compound 'Y' with sweet fragrance is formed. This compound is called methyl ethanoate, and its molecular formula is CH3COOCH3.
The formula with its functional group is represented as:
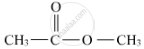
(b) The chemical equation of the above stated reaction is as follows:
`CH_3 COOH + C_2 H_5 OH` 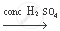 `CH_3 COOC_2 H_5 + H_2O`
`CH_3 COOC_2 H_5 + H_2O`
APPEARS IN
RELATED QUESTIONS
In an experiment to study the properties of ethanoic acid, a student takes about 3 mL of ethanoic acid in a dry test tube. He adds an equal amount of distilled water to it and shakes the test tube well. After some time he is likely to observe that
(A) a colloid is formed in the test tube.
(B) the ethanoic acid dissolves readily in water.
(C) the solution becomes light orange.
(D) water floats over the surface of ethanoic acid.
Complete the following chemical equations:
C2H5OH+CH3COOH`("conc."H_2SO_4)/`>
A gas is liberated immediately with a brisk effervescence, when you add acetic acid to sodium hydrogen carbonate powder in a test tube. Name the gas and describe the test that confirms the identity of the gas.
Complete the following chemical equations :CH3COOH + Na2CO3 →
Complete the following chemical equations: C2H5OH+O2→
Describe a test for carboxylic acid.
Vinegar is a solution of about:
(a) 5 to 8 per cent ethanoic acid in alcohol
(b) 5 to 8 per cent ethanoic acid in water
(c) 50 to 80 per cent ethanoic acid in water
(d) 50 to 80 per cent ethanoic acid in alcohol
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is a constituent of vinegar?(e) Which compound is a constituent of vinegar?
A student puts a drop of acetic acid first on a blue litmus paper and then on a red litmus paper. He would observe that
(A) the red litmus paper turns colourless and there is no change in the blue litmus paper.
(B) the red litmus paper turns blue and the blue litmus paper turns red.
(C) there is no change in the red litmus paper and the blue litmus paper turns red.
(D) there is no change in the blue litmus paper and the red litmus paper turns blue.
On adding 2 mL acetic acid to 2 mL of water in a test tube, it was observed that
(A) a clear and transparent solution is formed
(B) a white precipitate is formed almost immediately
(C) two separate layers were formed
(D) a colourless and odourless gas is evolved
Explain the following term with example.
Reduction
Fill in the blank with appropriate word/words.
Vinegar is a solution of about ________per cent ________in water.
Fill in the blank with appropriate word/words.
Esterification is the reaction between carboxylic acid and ______ in presence of ____
Fill in the blank with appropriate word/words.
Denatured alcohol is a mixture of _____ and _______
Choose the correct answer:
Select the acid which contains four hydrozen atoms in it.
Rewrite the following statement by selecting the correct option.
The formula of ethanoic acid is _____________.
Write the molecular formula of the given compound.
Sodium ethanoate
How is ethanol to ethanoic acid an oxidation reaction different from the reaction in which ethanol burns in the presence of oxygen?
Observe the diagram given below and answer the questions:

- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
