Advertisements
Advertisements
Question
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\ce{H2C = CH - CH - CH2 - CH2 - CH3}\\
|\phantom{..........}\\
\ce{OH}\phantom{........}
\end{array}\]
Advertisements
Solution
Hex-1-en-3-ol
APPEARS IN
RELATED QUESTIONS
What is metamerism?
Write IUPAC name of the following compound:
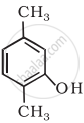
Write IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
How do you convert the Ethanal to Propanone
Propanoic acid to ethylamine.
Write structural formulae for 1-Ethylcyclohexanol.
Write structural formula for pentane-1,4-diol.
Write IUPAC names of the following

Which of the following compounds is oxidised to prepare methyl ethyl ketone?
n-Propyl alcohol and isopropyl alcohol can be chemically distinguished by which reagent?
Which of the following is most acidic?
Which of the following gives a positive iodoform test?
The correct acidic strength order of the following is:

(I)
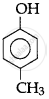
(II)

(III)
Explain why p-nitrophenol is more acidic than phenol.
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) |  |
(a) Hydroquinone |
| (ii) |  |
(b) Phenetole |
| (iii) | 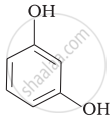 |
(c) Catechol |
| (iv) |  |
(d) o-Cresol |
| (v) |  |
(e) guinone |
| (vi) | 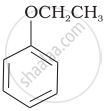 |
(f) Resorcinol |
| (g) Anisole |
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write the IUPAC name.
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C -CH3}\\
\phantom{.}|\phantom{......}|\phantom{......}|\\
\phantom{....}\ce{CH3\phantom{...}\ce{OH}\phantom{...}\ce{CH3}}\
\end{array}\]
