Advertisements
Advertisements
Question
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
Advertisements
Solution
i. The isomeric alcohols with molecular formula C5H12O are –
(a) \[\ce{\underset{Pentanol (1^\circ)}{CH3 - CH2 - CH2 - CH2 - CH2 - OH}}\]
(b) \[\begin{array}{cc}
\ce{CH3 - CH2 - CH2 - \overset{∗}{C}H - CH3}\\
\phantom{............}|\\
\phantom{...............}\ce{\underset{Pentan-2-ol (2^\circ)}{OH}}\
\end{array}\]
(c) \[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CH2 - CH3}\\
|\phantom{..}\\
\phantom{..}\ce{\underset{Pentan-3-ol (2^\circ)}{OH}}\
\end{array}\]
(d) \[\begin{array}{cc}
\ce{H3C - H2C - H\overset{∗}{C} - CH2OH}\\
\phantom{.....}|\\
\phantom{.........}\ce{\underset{2-Methylbutan-1-ol (1^\circ)}{CH3}}\
\end{array}\]
(e) \[\begin{array}{cc}
\ce{CH3}\phantom{............}\\
|\phantom{...............}\\
\ce{\underset{3-Methylbutan-1-ol (1^\circ)}{CH3 - CH - CH2 - CH2 - OH}}\\
\end{array}\]
(f) \[\begin{array}{cc}
\ce{CH3}\phantom{....}\\
|\phantom{.......}\\
\ce{CH3 - C - CH2 - CH3}\\
|\phantom{.......}\\
\ce{\underset{2-Methylbutan-2-ol (3^\circ)}{OH}}\phantom{....}\
\end{array}\]
(g) \[\begin{array}{cc}
\ce{CH3}\phantom{...}\\
|\phantom{......}\\
\ce{CH3 - C - CH2 - OH}\\
|\phantom{......}\\
\ce{\underset{2, 2-Dimethylpropan-1-ol (1^\circ)}{CH3}}\phantom{..}\
\end{array}\]
(h) \[\begin{array}{cc}
\phantom{.}\ce{CH3}\phantom{...}\ce{OH}\\
|\phantom{......}|\phantom{..}\\
\ce{\underset{3-Methylbutan-2-ol (2^\circ)}{CH3 - CH - \underset{∗}{C}H - CH3}}\
\end{array}\]
Isomers (b), (d) and (h) contain chiral carbon atoms; thus, they exhibit enantiomerism.
ii. Isomers (a), (d), (e) and (g) are primary alcohols.
Isomers (b), (c) and (h) are secondary alcohols.
Isomer (f) is a tertiary alcohol.
APPEARS IN
RELATED QUESTIONS
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\ce{H2C = CH - CH - CH2 - CH2 - CH3}\\
|\phantom{..........}\\
\ce{OH}\phantom{........}
\end{array}\]
Write IUPAC name of the following compound:
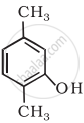
Write IUPAC name of the following compound:
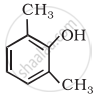
Give IUPAC name of the following ether:
\[\begin{array}{cc}
\ce{C2H5OCH2 - CH - CH3}\\
\phantom{.....}|\\
\phantom{.......}\ce{CH3}
\end{array}\]
Give IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Ethylidene dichloride when boiled with aqueous solution of NaOH yields _______.
(A) formaldehyde
(B) acetaldehyde
(C) acetone
(D) ethyl methyl ketone
What is the action of hot HI on it?
In the dehydration of alcohols to alkenes by heating with concentrated sulphuric acid, the initiation step is:
(1) formation of carbonation
(2) formation of an ester
(3) protonation of the alcohol molecule
(4) elimination of water
Give IUPAC names of the following compound:

Butane-2-ol is ____________.
Cresol has ____________.
Which of the following reagents can be used to oxidise primary alcohols to aldehydes?
(i) \[\ce{CrO3}\] in anhydrous medium.
(ii) \[\ce{KMnO4}\] in acidic medium.
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
Explain why p-nitrophenol is more acidic than phenol.
Match the starting materials given in Column I with the products formed by these (Column II) in the reaction with HI.
| Column I | Column II | ||
| (i) | CH3—O—CH3 | (a) |  |
| (ii) | \[\begin{array}{cc} \ce{CH3}\phantom{..................}\\ \backslash\phantom{.............}\\ \ce{CH-O-CH3}\\ /\phantom{..............}\\ \ce{CH3}\phantom{..................} \end{array}\] |
(b) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-I + CH3OH}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
| (iii) | \[\begin{array}{cc} \ce{CH3}\phantom{.}\\ |\phantom{....}\\ \ce{H3C-C-O-CH3}\\ |\phantom{....}\\ \ce{CH3}\phantom{..} \end{array}\] |
(c) | 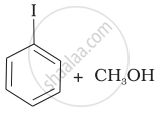 |
| (iv) |  |
(d) | CH3—OH + CH3—I |
| (e) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-OH + CH3I}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
||
| (f) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-I + CH3OH}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
||
| (g) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-OH + CH3I}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
How can phenol be converted to aspirin?
Draw structure of the following compound.
2. 5-Diethylphenol
Draw structure of the following compound.
2-Methoxypropane
Draw structure of the following compound.
Prop-2-en-1-ol
Write structural formulae for:
p-Nitrophenol
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.....}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{.}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
