Advertisements
Advertisements
Questions
Write the equation involved in the following reaction:
Reimer-Tiemann reaction
Explain the following with an example.
Reimer-Tiemann reaction
Write a chemical equation to illustrate the following name reaction:
Reimer-Tiemann reaction
Advertisements
Solution 1
On treating phenol with chloroform in the presence of sodium hydroxide, a -CHO group is introduced at the ortho position of the benzene ring. This reaction is known as the Reimer-Tiemann reaction. The intermediate substituted benzal chloride is hydrolysed in the presence of alkali to produce salicylaldehyde.

Solution 2
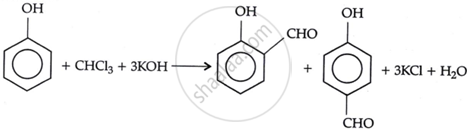
Solution 3
Phenol reacts with sodium hydroxide (or KOH) and chloroform in this reaction. 4-hydroxybenzaldehyde is a minor product, while salicylaldehyde is the primary product. Steam distillation can be used to separate it from the p-isomer. Intramolecular hydrogen bonding is responsible for the stability of the p-isomer.

Notes
Students should refer to the answers according to their questions and provide marks.
APPEARS IN
RELATED QUESTIONS
Give simple chemical tests to distinguish between the following pairs of compounds: Benzoic acid and Phenol
Write the structures of A, B, C, D and E in the following reactions:

Write the equation involved in the following reaction:
Kolbe’s reaction
While separating a mixture of ortho and para nitrophenols by steam distillation, name the isomer which will be steam volatile. Give reason.
Write the reaction involved in the following:
Friedal-Crafts Alkylation of Phenol
When phenol is treated with excess bromine water, it gives:
On distilling phenol with Zn dust, one gets:
The electrophile involved in Reimer-Tiemann reaction of phenol with CHCl3 in presence of NaOH:

Which of the following species can act as the strongest base?
Which of the following are used to convert \[\ce{RCHO}\] into \[\ce{RCH2OH}\]?
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
(iv) Reaction with \[\ce{RMgX}\] followed by hydrolysis
Out of o-nitrophenol and p-nitrophenol, which is more volatile? Explain.
Which of the following is not aromatic?
Write the equations for the following reaction:
Phenol is treated with chloroform in the presence of NaOH
Write the name of the reaction, structure and IUPAC name of the product formed when:
Phenol reacts with CHCl3 in the presence of NaOH followed by hydrolysis.
Convert the following by giving a chemical equation:
Phenol to salicylaldehyde.
