Advertisements
Advertisements
Question
Which of the following species can act as the strongest base?
Options
ΘOH
ΘOR
ΘOC6H5
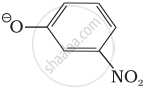
MCQ
Advertisements
Solution
ΘOR
Explanation:
Weakest acid has the strongest conjugate base. Since R-OH is the weakest acid, therefore, RO is the strongest base.
shaalaa.com
Is there an error in this question or solution?
