Advertisements
Advertisements
Question
Which of the following are used to convert \[\ce{RCHO}\] into \[\ce{RCH2OH}\]?
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
(iv) Reaction with \[\ce{RMgX}\] followed by hydrolysis
Advertisements
Solution
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
Explanation:
Aldehydes and ketones are reduced to the corresponding alcohols by addition of hydrogen in the presence of catalysts (catalytic hydrogenation. It is also prepared by treating aldehydes and ketones with sodium borohydride \[\ce{(NaBH4)}\] or lithium aluminium hydride \[\ce{(LiAlH4)}\].
APPEARS IN
RELATED QUESTIONS
Write the final product(s) in each of the following reactions:

Write the main product(s) in each of the following reactions:

While separating a mixture of ortho and para nitrophenols by steam distillation, name the isomer which will be steam volatile. Give reason.
Explain the following with an example.
Kolbe’s reaction.
When phenol is heated with CHCl3 and alcoholic KOH when salicylaldehyde is produced. This reaction is known as ____________.
On distilling phenol with Zn dust, one gets:
Which of the following species can act as the strongest base?
\[\ce{C2H5OH + SOCl2 ->[Pyridine] C2H5Cl + SO2 + HCl}\]
The above reaction is known as:
Phenol does not undergo nucleophilic substitution reaction easily due to ______.
Which of the following compounds is aromatic alcohol?
| (A) |  |
| (B) | 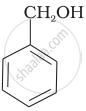 |
| (C) |  |
| (D) |  |
Which of the following are benzylic alcohols?
(i) \[\ce{C6H5 - CH2 - CH2OH}\]
(ii) \[\ce{C6H5 - CH2OH}\]
(iii) \[\begin{array}{cc}
\ce{C6H5 - CH - OH}\\
\phantom{}|\phantom{.}\\
\phantom{..}\ce{CH3}\phantom{}
\end{array}\]
(iv) \[\begin{array}{cc}
\ce{C6H5 - CH2 - CH - OH}\\
\phantom{.......}|\phantom{}\\
\phantom{.........}\ce{CH3}\phantom{}
\end{array}\]
Convert the following:
Phenol to N-phenylethanamide.
Which of the following is not aromatic?
Attacking species in nitration of benzene in presence of fuming HNO3 is
For the pair phenol and cyclohexanol, answer the following:
Give one chemical test to distinguish between the two.
