Advertisements
Advertisements
Question
Distinguish between Homolysis and heterolysis.
Advertisements
Solution
| No. | Homolysis (Homolytic fission) | Heterolysis (Heterolytic fission) |
| 1. | The symmetrical breaking of a covalent bond in which each departing atom takes one electron from the bonding pair is called as homolytic fission. | The unsymmetrical breaking of a covalent bond in which one of the departing atoms retains the bonding pair is called heterolytic fission. |
| 2. | In this type of fission, the formation of free radicals (uncharged species) bearing unpaired electrons takes place. | In this type of fission, the formation of charged species called ions, like carbocation or carbonium ion takes place. |
| 3. |
The covalent bond between two atoms of the same element or two atoms having nearly the same electronegativity breaks in this manner.
|
The covalent bond between two atoms of the different elements or two atoms having different electronegativity values breaks in this manner.
|
| 4. | This takes place favourably in a nonpolar solvent. | This takes place favourably in a polar solvent. |
| 5. | Generally, reaction takes place at high temperature or in presence of UV light or peroxides. | Heterolysis takes place in solutions (polar condition). |
APPEARS IN
RELATED QUESTIONS
Draw a resonance structure of the following:
Benzaldehyde
Draw a resonance structure of the following:
Acetate ion
Distinguish between Inductive effect and resonance effect.
Write true or false. Correct the false statement.
Aniline is a heterocyclic compound.
The correct IUPAC name of the compound 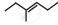 is ______.
is ______.
Which among the following is a set of nucleophiles?
Which of the following is TRUE for homolytic fission?
Identify the reagent used in the following reaction:
\[\ce{CH3 - CH2 - Br ->[?] CH3 - CH2 - OH}\]
The best reagent for the following conversion is:

Which of the following alkyl groups shows maximum positive inductive effect?
Which of the following is the most unstable carbocation?
Which of the following alkyl groups shows least positive inductive effect?
Which of the following is NOT an electrophile?
Which of the following statements is not correct?
The most probable product in the reaction given below is:

Which element among the following does form pπ - pπ multiple bonds?
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
CH2 = CH - CH2 - CH3
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α - carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H -CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogens.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]


