Advertisements
Advertisements
Question
Distinguish between Carbocation and carbanion.
Advertisements
Solution
| No. | Carbocation | Carbanion |
| 1. | It is a species in which carbon carries a positive charge. | It is a species in which carbon carries a negative charge. |
| 2. | Positively charged carbon is sp2 hybridized. | Negatively charged carbon is sp3/sp2 hybridized. |
| 3. | It is electron-deficient. | It is electron-rich |
| 4. | It has a planar geometry. | It has a tetrahedral geometry. |
| 5. | e.g. tert-Butyl carbocation, (CH3)3C+ | Methyl carbanion |
APPEARS IN
RELATED QUESTIONS
Write bond-line formulae and condensed formulae for the following compound.
3-methyloctane
Write bond-line formulae and condensed formulae for the following compound.
octa-1,4-diene
Write bond-line formulae and condensed formulae for the following compound.
Methoxyethane
Write the structural formulae for the following name and also write the correct IUPAC name for it.
2,2,3-trimethylpentan-4-ol
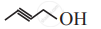
The following structure can be represented as:
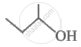
What is the molar mass of a compound represented below?
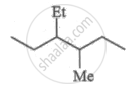
Write dash formulae for the following bond line formulae.
![]()
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\phantom{......}\ce{H}\phantom{...} \ce{H} \phantom{...}\ce{H}\phantom{....} \ce{O}\\
\phantom{......}|\phantom{....}|\phantom{....}|\phantom{....}// \\
\ce{H - C - C - C - C }\\
\phantom{......}|\phantom{....}|\phantom{....}|\phantom{.....}\backslash \\
\phantom{..........}\ce{H}\phantom{...} \ce{H} \phantom{...}\ce{H}\phantom{....} \ce{OH}\phantom{...}
\end{array}\]
Write bond line formulae and condensed formulae for the following compound.
2, 2, 4, 4- tetramethylpentane
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{}\\
\end{array}\]
Write dash formulae for the following bond line formulae.
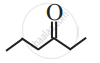
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\phantom{}\ce{H\phantom{...}H\phantom{...}H\phantom{...}H}\phantom{}\\
\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}\\
\ce{H - C - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{}\ce{H\phantom{...}H\phantom{...}H\phantom{...}H}\phantom{}
\end{array}\]
Write the structural formulae for the following name and also write the correct IUPAC name for them.
2,2,3-trimethylpentan-4-0l
Write dash formulae for the following bond line formulae.
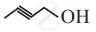
Write bond line formulae and condensed formulae for the following compound.
2,2,4,4-tetramethylpentane
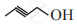
Write dash formulae for the following bond line formulae.
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\phantom{.......}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.....}\ce{O}\\
\phantom{.......}|\phantom{....}|\phantom{....}| \phantom{....}//\\
\ce{H - C - C - C - C}\\
\phantom{......}|\phantom{....}|\phantom{....}| \phantom{.....}\backslash\\
\phantom{........}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.....}\ce{OH}
\end{array}\]
Write bond line formulae and condensed formulae for the following compound.
2, 2, 4, 4- tetramethylpentane
Write the structural formulae for the following name and also write correct IUPAC name.
2,2,3-trimethylpentan-4-ol
Write the structural formulae for the following name and also write correct IUPAC name.
2,2,3-trimethylpentan-4-ol
Write bond-line formulae and condensed formulae for the following compound.
2, 2, 4, 4- tetramethylpentane
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\phantom{......}\ce{H\phantom{...}H\phantom{...}H\phantom{.....}O}\\
\phantom{......}|\phantom{....}|\phantom{....}|\phantom{....}//\\
\ce{H - C - C - C - C}\\
\phantom{......}|\phantom{....}|\phantom{....}|\phantom{.....}\backslash\\
\phantom{........}\ce{H\phantom{...}H\phantom{...}H\phantom{.....}OH}
\end{array}\]
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\phantom{.........}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.......}\ce{O}\\
\phantom{........}|\phantom{....}|\phantom{....}|\phantom{......}//\\
\ce{H - C - C - C - C}\\
\phantom{.......}|\phantom{....}|\phantom{....}|\phantom{......}\backslash\\\
\phantom{..........}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.......}\ce{OH}\\
\end{array}\]
Write bond line formula and condensed formula for the following compound.
2, 2, 4, 4-tetramethylpentane
Write bond line formulae and condensed formulae for the following compound.
2, 2, 4, 4- tetramethylpentane
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\ce{H\phantom{...}H\phantom{...}H\phantom{...}H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H\phantom{...}H\phantom{...}H\phantom{...}H}
\end{array}\]
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\phantom{.........}\ce{H\phantom{...}H\phantom{...}H}\phantom{......}\ce{O}\phantom{.}\\
\phantom{.........}|\phantom{....}|\phantom{....}|\phantom{.....}//\phantom{..}\\
\ce{H - C - C - C - C}\\
\phantom{...........}|\phantom{....}|\phantom{....}|\phantom{.....}\backslash\phantom{.....}\\
\phantom{..........}\ce{H\phantom{...}H\phantom{...}H}\phantom{.....}\ce{OH}\phantom{..}
\end{array}\]
Write dash formulae for the following bond line formulae.