Advertisements
Advertisements
Question
Choose the correct option.
The delocalization of electrons due to overlap between p orbital and sigma bond is called _______.
Options
inductive effect
electronic effect
hyperconjugation
resonance
Advertisements
Solution
The delocalization of electrons due to overlap between p orbital and sigma bond is called hyperconjugation.
APPEARS IN
RELATED QUESTIONS
Find out the most stable species from the following. Justify.
`bar"C""H"_3, bar"C""H"_2"Br", bar"C""Br"_3`
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH_3 - CH_2 - \overset{⊕}{C}H-CH_2 - CH_3}\]
Draw a resonance structure of the following:
Phenol
Draw a resonance structure of the following:
Acetate ion
Distinguish between Inductive effect and resonance effect.
Write true or false. Correct the false statement.
Aniline is a heterocyclic compound.
Choose the correct option.
Hyperconjugation involves overlap of ______ orbitals.
The correct IUPAC name of the compound 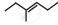 is ______.
is ______.
Predict the order of reactivity of the following compounds by SNl reaction mechanism.
\[\ce{\underset{\text{(I)}}{C6H5CH(C6H5)Cl}}\]
\[\ce{\underset{\text{(II)}}{C6H5CH2Cl}}\]
\[\ce{\underset{\text{(III)}}{C6H5C(CH3)(C6H5)Cl}}\]
Which of the following compound is highly reactive towards HCN?
Which of the following is the strongest nucleophile?
Which of the following is TRUE for homolytic fission?
The best reagent for the following conversion is:

The overlap of σ-p orbitals is called ____________.
Which of the following is the most unstable carbocation?
IUPAC name of ![]() is ______.
is ______.
How many pi bonds and sigma bonds are present in following molecule?
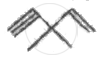
Which element among the following does form pπ - pπ multiple bonds?
Identify the group that exerts electron withdrawing resonance effect.
Identify the functional group that has an electron-donating inductive effect.
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogens.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
