Advertisements
Advertisements
Question
Identify the α-carbon in the following species and give the total number of α-hydrogens.
\[\ce{CH2 = CH - CH2 - CH3}\]
Advertisements
Solution
\[\ce{\overset{1}{C}H2 = \overset{2}{C}H - \overset{3}{\underset{\alpha}{C}}H2 - \overset{4}{C}H3}\]
The C-3 carbon atom, that is, the C-atom next to \[\ce{(H2C = CH-)}\] is an α-C atom.
Thus, the structure contains 2 α-H atoms.
APPEARS IN
RELATED QUESTIONS
Find out the most stable species from the following. Justify
`dot"CH","CH"_3-dot"CH" - "CH"_3,` \[\begin{array}{cc}\ce{CH3 -\dot{C} - CH3}\\
|\phantom{.}\\\phantom{..}\ce{CH3}\end{array}\]
Find out the most stable species from the following. Justify.
\[\ce{\overset{+}{C}H3, \overset{+}{C}H2Cl, \overset{+}{C}Cl3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen.

Draw a resonance structure of the following:
Buta-1,3-diene
Distinguish between Inductive effect and resonance effect.
Write true or false. Correct the false statement.
Heterolytic fission results in the formation of free radicals.
Choose the correct option.
Which of the following statements are true with respect to electronic displacement in a covalent bond?
a. Inductive effect operates through π bond
b. Resonance effect operates through σ bond
c. Inductive effect operates through σ bond
d. Resonance effect operates through π bond
Choose the correct option.
Hyperconjugation involves overlap of ______ orbitals.
The correct IUPAC name of the compound 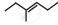 is ______.
is ______.
Choose the correct option.
The delocalization of electrons due to overlap between p orbital and sigma bond is called _______.
Predict the order of reactivity of the following compounds by SNl reaction mechanism.
\[\ce{\underset{\text{(I)}}{C6H5CH(C6H5)Cl}}\]
\[\ce{\underset{\text{(II)}}{C6H5CH2Cl}}\]
\[\ce{\underset{\text{(III)}}{C6H5C(CH3)(C6H5)Cl}}\]
Which of the following shows positive resonance (+R) effect?
Which of the following alkyl groups shows maximum positive inductive effect?
The most unstable free radical among the following is:
IUPAC name of ![]() is ______.
is ______.
Which of the following alkyl groups shows least positive inductive effect?
Which of the following represents a set of nucleophiles?
Which of the following statements is not correct?
Identify the functional group that has an electron-donating inductive effect.
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
CH2 = CH - CH2 - CH3
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α - carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H -CH2 - CH3}\]
Which of the following groups exhibits a +I effect?
