Advertisements
Advertisements
Question
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Advertisements
Solution
- Buna-S:
It has mechanical strength and has abrasion resistance. Hence, it is used in tyre industry. - Neoprene:
- Neoprene is particularly resistant to petroleum, vegetable oils, light as well as heat.
- Hence, it is used in making hose pipes for the transport of gasoline and making gaskets.
- It is used for manufacturing insulator cable, jackets, belts for power transmission and conveying.
APPEARS IN
RELATED QUESTIONS
Write the chemical reaction to prepare novolac polymer.
Write the structure of melamine.
Write the structures of the monomers used for getting the following polymers
Melamine – formaldehyde polymer
Answer the following in one sentence.
Identify 'A' in the following reaction:
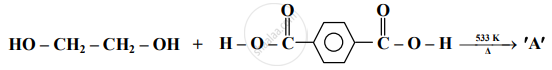
Answer the following in one sentence.
Define the term: Vulcanization
Answer the following.
Write the reaction of the formation of Nylon 6.
Answer the following.
Write structure of natural rubber and neoprene rubber along with the name and structure of their monomers.
Answer the following.
Draw the structures of polymers formed from the following monomers
H2N–(CH2)5 – COOH
Name and draw structure of the repeating unit in natural rubber.
Answer the following.
Write name and formula of raw material from which bakelite is made.
Identify condensation polymers and addition polymers from the following.
Attempt the following:
Explain the vulcanisation of rubber. Which vulcanizing agents are used for the following synthetic rubber?
a. Neoprene
b. Buna-N
Write the reaction involved in the formation of bakelite.
Write the structure of isoprene and the polymer obtained from it.
Monomer of natural rubber is __________
Write a chemical reaction for the preparation of the following polymer.
polyacrylonitrile
Write chemical reaction for preparation of the following.
Buna-S
Explain vulcanization of rubber.
Which among the following polymers is obtained from styrene and 1-3-butadiene?
Which among the following polymers is used for making handles of cooker?
How many isoprene units are present in abscisic acid?
Identify the CORRECT statement regarding the following polymer.
\[\begin{array}{cc}
\phantom{....}\ce{O}\phantom{............}\ce{O}\phantom{...................}\ce{H}\phantom{.....}\\
\phantom{....}||\phantom{.............}||\phantom{...................}|\phantom{......}\\
\ce{-[C - (CH2)4 - C - NH - (CH2)6 - N -]_{{n}}}
\end{array}\]
The INCORRECT match for the polymer with its application is:
Select the CORRECT match for both the polymers.
\[\ce{{n} CH2 = CH2 ->[333 K - 343 K][6 - 7 atm, catalyst] X}\]
Which of the following is CORRECT about polymer 'X'?
Which of the following polymers is obtained from chloroprene?
Which among the following polymers can NOT be remoulded?
Which among the following catalysts is used in the preparation of dacron?
Which among the following monomers is used to prepare Teflon?
Which of the following polymer is used to make blankets?
Which among the following is an example of addition polymer?
Match the polymers given in Column I with the type of linkage present in them given in Column II.
| Column I | Column II |
| (i) Terylene | (a) Glycosidic linkage |
| (ii) Nylon | (b) Ester linkage |
| (iii) Cellulose | (c) Phosphodiester linkage |
| (iv) Protein | (d) Amide linkage |
| v) RNA |
Which of the following is not a copolymer?
Which of the following polymer has ester linkage?
Which one of the following polymers are prepared by addition polymerization?
Which of the following polymer is used for manufacturing of buckets, dustbins, pipes, etc?
Which among the following polymers has high tensile strength and is used to obtain tyre cords?
The monomer of natural rubber is ______.
Answer the following.
Write the structure of isoprene and the polymer obtained from it.
Name and draw the structure of the repeating unit in natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the name of the monomer of natural rubber.
Write the structure and name of the monomer of Nylon-6.
Write the structure and name of monomer of Natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw the structure of the repeating unit in natural rubber.
Which from following statements is NOT true regarding neoprene?
Which among the following is a monomer of natural rubber?
