Advertisements
Advertisements
Questions
Write chemical reaction for preparation of the following.
Buna-S
Write chemical reaction for the preparation of the following polymer.
SBR
Write an equation for the preparation of the following:
Buna-S
Give the chemical equations for the preparation of Buna-S.
How is Buna-S synthesised? Give a chemical equation.
Write the chemical equation to represent the preparation of Buna-N.
Advertisements
Solution 1
Chemical reaction for preparation of Buna-S:

Solution 2
It is made by polymerizing a 75% butadiene and 25% styrene mixture in an emulsion system at 0°C with a peroxide catalyst present.

RELATED QUESTIONS
Write any ‘two’ uses of terylene.
Explain the following term: Homopolymers
Write the formulae of the raw materials used for preparation of Dextran.
Write the structure of melamine.
Answer the following in one sentence.
Identify 'A' in the following reaction:
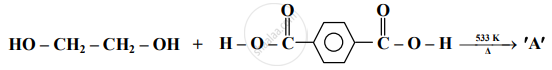
Answer the following in one sentence.
Define the term: Vulcanization
Answer the following in one sentence.
What type of intermolecular force leads to high-density polymer?
Answer the following in one sentence.
Identify thermoplastic and thermosetting plastic from the following:
- PET
- Urea formaldehyde resin
- Polythene
- Phenol formaldehyde resin
Answer the following.
Write structure of natural rubber and neoprene rubber along with the name and structure of their monomers.
Answer the following.
Name the polymer type in which following linkage is present.
\[\begin{array}{cc}\ce{- C - O -}\\||\phantom{.....}\\
\ce{O\phantom{.....}}\end{array}\]
Draw the structures of polymers formed from the following monomers
\[\ce{n HOOC–R–COOH + n HO–R'–OH}\]
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Write the reaction involved in the formation of:
Bakelite
Write the structure of isoprene and the polymer obtained from it.
Write the name of the catalyst used for preparation of high density polythene polymer.
Monomer used for preparation of polyacrylonitrile is _____________
Write chemical reactions for the preparation of high-density polythene.
Write two uses and two properties of polythene.
Explain vulcanization of rubber.
Define rubber.
Explain the reactions involved in the preparation of viscose rayon.
How many isoprene units are present in abscisic acid?
Which among the following polymers can NOT be remoulded?
Which of the following catalysts is used in preparation of terylene?
Identify the catalyst used in the manufacture of high density polythene.
Which of the following polymers is prepared by using phenol?
Which among the following polymers is an example of addition polymer?
Which of the following polymers is used as insulation for cables?
Which among the following is an example of addition polymer?
Phenol and formaldehyde undergo condensation to give a polymar (A) which on heating with formaldehyde gives a thermosetting polymer (B). Name the polymers. Write the reactions involved in the formation of (A). What is the structural difference between two polymers?
Which of the following polymer has ester linkage?
Which of the following is an example of polyester?
Polymer used in bullet proof glass is ______.
Answer the following.
Write the structure of isoprene and the polymer obtained from it.
Answer the following.
Write the structure of isoprene and the polymer obtained from it.
Write the preparation of viscose rayon.
Write the structure of isoprene and the polymer obtained from it.
Match the following pairs:
| Polymer | Monomer | ||
| (i) | Teflon | (a) | CH2 = CH2 |
| (ii) | PVC | (b) | CF2 = CF2 |
| (iii) | Natural rubber | (c) | Isoprene |
| (iv) | Polythene | (d) | H2C=CHCl |
The monomer used in preparation of teflon is ______.
Write the structure and name of monomer of Natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Name and draw the structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
