Advertisements
Advertisements
Question
Write chemical reactions for the preparation of high-density polythene.
Advertisements
Solution
Chemical reaction for the preparation of high-density polythene (HDP) is as follows:

APPEARS IN
RELATED QUESTIONS
Write the chemical reaction to prepare novolac polymer.
Explain the following term: Homopolymers
The Zieglar-Natta catalyst is used in the preparation of _______.
(A) LDPE
(B) PHBV
(C) PAN
(D) HDPE
Write the structures of the monomers used for getting the following polymers
Melamine – formaldehyde polymer
Answer the following in one sentence.
Define the term: Vulcanization
Answer the following.
Write name and formula of raw material from which bakelite is made.
Identify condensation polymers and addition polymers from the following.
\[\begin{array}{cc}\ce{-(CH2 - CH -)_{n}}\\
\phantom{....}|\\\ce{\phantom{.......}C6H5}
\end{array}\]
Identify condensation polymers and addition polymers from the following.
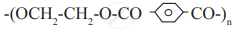
Write the reaction involved in the formation of bakelite.
Monomers ethylene glycol and terephthalic acid undergo condensation polymerization to give polymer called ___________
Write a chemical reaction for the preparation of the following polymer.
polyacrylonitrile
Write preparation of low density polythene.
Mention two uses of LDP.
Write chemical reaction for preparation of the following.
Buna-S
Write the name of one example of each polymer in which following repeating units.
\[\begin{array}{cc}
\ce{(-CF2-CF2-), -[NH-(CH2)5-CO] -, -(CH2-CH-), (-CH2-CH2-)}\\
\phantom{............................}|\\
\phantom{..............................}\ce{CN}
\end{array}\]
Which of the following is used as a substitute for wool?
The following structure represents the polymer:
\[\begin{array}{cc}
\ce{[-C-CH2-NH-C-(-CH2)5 NH -]_{{n}}}\\
\phantom{}||\phantom{.............}||\phantom{................}\\
\phantom{}\ce{O}\phantom{.............}\ce{O}\phantom{................}
\end{array}\]
Which among the following polymers is used for making handles of cooker?
Select the CORRECT match for both the polymers.
Novolac is obtained from ____________.
Identify the polymer obtained by polymerization of n moles of acrylonitrile.
Which among the following polymers can NOT be remoulded?
Which among the following polymers is obtained from CH2 = CH – CN by polymerisation?
Identify the monomers used in the preparation of Novolac.
Which among the following monomers is used to prepare Teflon?
Which among the following polymers is an example of addition polymer?
Which of the following polymers is used as insulation for cables?
Which among the following is an example of addition polymer?
Which of the following is not a semisynthetic polymer?
The commercial name of polyacrylonitrile is ______.
Which of the following polymers, need atleast one diene monomer for their preparation?
(i) Dacron
(ii) Buna-S
(iii) Neoprene
(iv) Novolac
Match the polymers given in Column I with their repeating units given in Column II.
| Column I | Column II |
| (i) Acrilan |
(a) \[\begin{array}{cc} |
| (ii) Polystyrene | (b) \[\begin{array}{cc} \ce{Cl}\phantom{.......}\\ |\phantom{........}\\ \phantom{}\ce{-(CH2 - C = CH - CH2)\underset{n}{-}} \end{array}\] |
| (iii) Neoprene | (c) \[\begin{array}{cc} \phantom{................................}\ce{CN}\\ \phantom{..............................}|\\ \ce{-(CH2 - CH = CH - CH2 - CH2 - CH)\underset{n}{-}} \end{array}\] |
| (iv) Novolac | (d) \[\begin{array}{cc} \ce{-(CH2 - CH)\underset{n}{-}}\\ \phantom{.....}|\\ \phantom{.......}\ce{CN} \end{array}\] |
| (v) Buna—N | (e)  |
| (f) \[\begin{array}{cc} \ce{-(CH2 - CH)\underset{n}{-}}\\ \phantom{.....}|\\ \phantom{......}\ce{Cl} \end{array}\] |
Which of the following is not a copolymer?
Which of the following polymer has ester linkage?
Which one of the following polymers are prepared by addition polymerization?
Nylon threads are made of ______.
Polymer used in bullet proof glass is ______.
Which of the following polymer is used for manufacturing of buckets, dustbins, pipes, etc?
Answer the following.
Write the structure of isoprene and the polymer obtained from it.
Answer the following.
Write the structure of isoprene and the polymer obtained from it.
Name the compound which reacts with formaldehyde to produce ethyl alcohol.
Write the name of the monomer of natural rubber.
The monomer used in preparation of teflon is ______.
Write the structure and name of monomer of Natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
