Advertisements
Advertisements
Question
Define rubber.
Advertisements
Solution
Polymers which are elastic in nature are called rubber.
APPEARS IN
RELATED QUESTIONS
Bakelite is the polymer of:
(a) Benzaldchyde and phenol
(b) Acetaldehyde and phenol
(c) Formaldehyde and phenol
(d) Formaldehyde and benzyl alcohol
Based on molecular forces, what type of polymer is neoprene?
Answer the following in one sentence.
Identify 'A' in the following reaction:
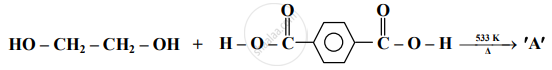
Answer the following in one sentence.
What type of intermolecular force leads to high-density polymer?
Name and draw structure of the repeating unit in natural rubber.
Write the name of the catalyst used for preparation of high density polythene polymer.
Monomer used for preparation of polyacrylonitrile is _____________
Monomer of natural rubber is __________
Write preparation of low density polythene.
Mention two uses of LDP.
Write the name of one example of each polymer in which following repeating units.
\[\begin{array}{cc}
\ce{(-CF2-CF2-), -[NH-(CH2)5-CO] -, -(CH2-CH-), (-CH2-CH2-)}\\
\phantom{............................}|\\
\phantom{..............................}\ce{CN}
\end{array}\]
Write chemical reactions for the preparation of high-density polythene.
Explain the reactions involved in the preparation of viscose rayon.
Which among the following polymers is obtained from styrene and 1-3-butadiene?
Which among the following polymers is used for making handles of cooker?
Identify additional polymers from the following.
I. \[\begin{array}{cc}
\ce{-(CH2 - CH -)_{{n}}}\\
\phantom{....}|\\
\phantom{.......}\ce{C6H5}
\end{array}\]
II. \[\ce{-(CH2 - CH = CH - CH2 -)_{{n}}}\]
III. \[\ce{-(CO(CH2)4 - CONH(CH2)6NH -)_{{n}}}\]
IV.
![]()
How many isoprene units are present in abscisic acid?
Identify the CORRECT statement regarding the following polymer.
\[\begin{array}{cc}
\phantom{....}\ce{O}\phantom{............}\ce{O}\phantom{...................}\ce{H}\phantom{.....}\\
\phantom{....}||\phantom{.............}||\phantom{...................}|\phantom{......}\\
\ce{-[C - (CH2)4 - C - NH - (CH2)6 - N -]_{{n}}}
\end{array}\]
Which of the following polymers is a heteropolymer?
Which of the following catalysts is used in preparation of terylene?
Identify the catalyst used in the manufacture of high density polythene.
Which among the following monomers is used to prepare Teflon?
Which among the following polymers is an example of addition polymer?
Which of the following pair of compounds is used as monomers for bakelite?
Which among the following polymers is used to manufacture chemical containers?
Identify addition polymer from the following.
Name the polymers used in laminated sheets and give the name of monomeric units involved in its formation.
Match the polymers given in Column I with the type of linkage present in them given in Column II.
| Column I | Column II |
| (i) Terylene | (a) Glycosidic linkage |
| (ii) Nylon | (b) Ester linkage |
| (iii) Cellulose | (c) Phosphodiester linkage |
| (iv) Protein | (d) Amide linkage |
| v) RNA |
Phenol and formaldehyde undergo condensation to give a polymar (A) which on heating with formaldehyde gives a thermosetting polymer (B). Name the polymers. Write the reactions involved in the formation of (A). What is the structural difference between two polymers?
Which of the following polymer has ester linkage?
Which of the following is an example of polyester?
Which of the following polymers is synthesized using a free radical polymerisation technique?
Which of the following polymers do not involve cross linkages?
Which of the following is a polymer of enzyme?
The monomer of natural rubber is ______.
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the preparation of viscose rayon.
Name and draw structure of the repeating unit in natural rubber.
The monomer used in preparation of teflon is ______.
Write the structure of isoprene and the polymer obtained from it.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Which among the following is a monomer of natural rubber?
