Advertisements
Advertisements
Question
Write preparation of low density polythene.
Advertisements
Solution
Preparation of low density polythene:
- LDP is prepared by polymerization of ethylene under high pressure (1000 – 2000 atm) and temperature (350 – 570 K) in presence of traces of O2 or peroxide as initiator.
\[\ce{{n} CH2 = CH2 ->[Traces of O2 or][peroxide at 350–570 K, 1000–2000 atm] LDP}\] - The mechanism of this reaction involves free radical addition and H-atom abstraction. The latter results in branching.
- Polymeric chains are loosely held due to branching and the polymer has low density.
APPEARS IN
RELATED QUESTIONS
Bakelite is the polymer of:
(a) Benzaldchyde and phenol
(b) Acetaldehyde and phenol
(c) Formaldehyde and phenol
(d) Formaldehyde and benzyl alcohol
The Zieglar-Natta catalyst is used in the preparation of _______.
(A) LDPE
(B) PHBV
(C) PAN
(D) HDPE
Write the structures of the monomers used for getting the following polymers
Melamine – formaldehyde polymer
Write the monomers of the following polymer :

Choose the correct option from the given alternatives.
Which of the following is made up of polyamides?
Answer the following in one sentence.
What type of intermolecular force leads to high-density polymer?
Answer the following.
Name the polymer type in which following linkage is present.
\[\begin{array}{cc}\ce{- C - O -}\\||\phantom{.....}\\
\ce{O\phantom{.....}}\end{array}\]
Identify condensation polymers and addition polymers from the following.
-(CH2 - CH = CH - CH2 -)n
Identify condensation polymers and addition polymers from the following.
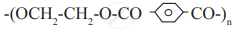
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Attempt the following:
Explain the vulcanisation of rubber. Which vulcanizing agents are used for the following synthetic rubber?
a. Neoprene
b. Buna-N
Attempt the following:
What is meant by LDP and HDP? Mention the basic difference between the same with suitable examples.
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Monomer used for preparation of polyacrylonitrile is _____________
Monomer of natural rubber is __________
Write the name of one example of each polymer in which following repeating units.
\[\begin{array}{cc}
\ce{(-CF2-CF2-), -[NH-(CH2)5-CO] -, -(CH2-CH-), (-CH2-CH2-)}\\
\phantom{............................}|\\
\phantom{..............................}\ce{CN}
\end{array}\]
Write the name and formulae of the monomers used for the preparation of dacron.
Explain vulcanization of rubber.
Define rubber.
Which of the following is used as a substitute for wool?
Identify the CORRECT statement regarding the following polymer.
\[\begin{array}{cc}
\phantom{....}\ce{O}\phantom{............}\ce{O}\phantom{...................}\ce{H}\phantom{.....}\\
\phantom{....}||\phantom{.............}||\phantom{...................}|\phantom{......}\\
\ce{-[C - (CH2)4 - C - NH - (CH2)6 - N -]_{{n}}}
\end{array}\]
The INCORRECT match for the polymer with its application is:
Novolac is obtained from ____________.
Which among the following polymers can NOT be remoulded?
Which of the following compounds is used to prepare orlon?
Which among the following monomers is used to prepare Teflon?
Which among the following polymers is an example of addition polymer?
Which among the following is an example of addition polymer?
Which of the following polymers, need atleast one diene monomer for their preparation?
(i) Dacron
(ii) Buna-S
(iii) Neoprene
(iv) Novolac
Match the polymers given in Column I with their repeating units given in Column II.
| Column I | Column II |
| (i) Acrilan |
(a) \[\begin{array}{cc} |
| (ii) Polystyrene | (b) \[\begin{array}{cc} \ce{Cl}\phantom{.......}\\ |\phantom{........}\\ \phantom{}\ce{-(CH2 - C = CH - CH2)\underset{n}{-}} \end{array}\] |
| (iii) Neoprene | (c) \[\begin{array}{cc} \phantom{................................}\ce{CN}\\ \phantom{..............................}|\\ \ce{-(CH2 - CH = CH - CH2 - CH2 - CH)\underset{n}{-}} \end{array}\] |
| (iv) Novolac | (d) \[\begin{array}{cc} \ce{-(CH2 - CH)\underset{n}{-}}\\ \phantom{.....}|\\ \phantom{.......}\ce{CN} \end{array}\] |
| (v) Buna—N | (e)  |
| (f) \[\begin{array}{cc} \ce{-(CH2 - CH)\underset{n}{-}}\\ \phantom{.....}|\\ \phantom{......}\ce{Cl} \end{array}\] |
Which of the following products is formed when benzaldehyde is treated with CH3MgBr and the addition product so obtained is subjected to acid hydrolysis?
Which one of the following polymers are prepared by addition polymerization?
Polymer used in bullet proof glass is ______.
Which of the following polymers do not involve cross linkages?
The catalyst used for the polymerisation of olefins is ______.
Which among the following polymers has high tensile strength and is used to obtain tyre cords?
The monomer of natural rubber is ______.
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Answer the following.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
The monomer used in preparation of teflon is ______.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Name and draw the structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
