Advertisements
Advertisements
Question
Answer in brief.
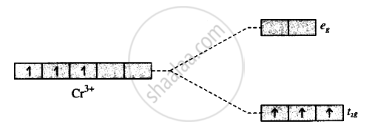
With the help of the crystal field, the energy-level diagram explains why the complex [Cr(en)3]3⊕ is coloured.
Advertisements
Solution
CFT:
ligands are treated as point charges interaction, but ligands & metal are purely electrostatic.
Metal has dorbital = dxy, dxz, dyz, dx2, dy2, dz2
When ligands approach metal, the degeneracy of metal is disturbed.
spit int 2 energy levels = t2g & eg
dxy, dxz, dxz
[Cr(en)3]+3 = en = ethylene diamine bidentate ligand
n = 3
`sqrt(n(n+2)) = sqrt(3(3+2))`
`sqrt15`
= 3.83 B.M
Paramagnetic :
d - d transition = [Cr(en)3]+3 = coloured property
APPEARS IN
RELATED QUESTIONS
On the basis of CFT predict the number of unpaired electrons in [CrF6]3-.
Answer in brief.
[CoCl4]2− is a tetrahedral complex. Draw its box orbital diagram. State which orbitals participate in hybridization.
Answer in brief.
What are strong field and weak field ligands ? Give one example of each.
Answer the following question.
Give valence bond description for the bonding in the complex [VCl4]-. Draw box diagrams for the free metal ion. Which hybrid orbitals are used by the metal? State the number of unpaired electrons.
Explain, why [Co(NH3)6]3+ ion is low spin? Calculate number of unpaired electrons and write the geometry of [Co(NH3)6]3+.
Chlorophyll and haemoglobin are complexes of ____________ respectively.
The number of unpaired electrons in the complex ion [CoF6]3− is ____________.
The INCORRECT match for complex with its geometry is:
The number of unpaired electrons in the complex ion [NiCl4]2− is ____________.
What is the value of primary valence of Co in CoCl3?
Identify the increasing order of effective magnetic moment of the following elements in their +2 oxidation state.
[Fe (Z = 26), Co (Z = 27), Ni (Z = 28), Cu (Z = 29)]
Which of the following types of square planar complexes can show geometrical isomerism [M = metal, a, b, = monodentate ligand]?
Explain the formation of [CoF6]3Θ complex with respect to
- Hybridisation
- Magnetic properties
- Inner/outer complex
- Geometry
Give VBT description of the bonding in a square planar complex [Cu(NH3) 4]2+. Show orbital diagrams for free metal ion and metal ion in the complex. Which hybrid orbitals are used by metal for bonding? State magnetic nature of the complex.
A compound forms a hep structure. Calculate the number of octahedral voids in 0.4 mol. (NA = 6.022 × 1023).
A compound forms a hep structure. Calculate the number of octahedral voids in 0.4 mol. (NA = 6.022 × 1023 )
Octahedral complex have ______ hybridisation.
Give the limitations of VBT.
Mention the type of hybridization in [Co(NH3)6]3+ complex.
Mention the number of unpaired electrons and geometry of the following complex:
\[\ce{[Ni(Cl)4]^{2-}}\]
Mention the number of unpaired electrons and geometry of the following complex:
\[\ce{[Ni(CN)4]^2-}\]
